��Ŀ����
2��̼��þˮ�������Ʊ�þ��Ʒ���м��壬��ҵ�ϴ�������±ˮ����Ҫ�ɷ�ΪMgCl2���л�ȡMgCO3•3H2O�ķ�����ͼ1����1��д���������̵����ӷ���ʽ��Mg2++CO2+2OH-+2H2O�TMgCO3•3H2O��
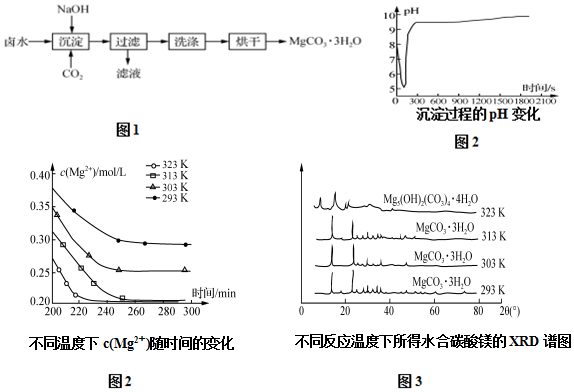
��2���������̵�pH��ʱ��ı仯��ͼ2��ʾ���������̵IJ���Ϊc ������ţ�
a����±ˮ�еμ�NaOH��Һ��ͬʱͨ��CO2
b����NaOH��Һ�еμ�±ˮ��ͬʱͨ��CO2
c����±ˮ��ͨ��CO2�����ͣ�Ȼ��μ�NaOH��Һ��ͬʱ����ͨ��CO2
d����NaOH��Һ��ͨ��CO2�����ͣ�Ȼ��μ�±ˮ��ͬʱ����ͨ��CO2
��3���������̵�c��Mg2+����ʱ��ı仯��ͼ3��ʾ����ͬ�¶������õ��ij���������ͼ4��ʾ����������Ӧѡ����¶�Ϊ313K�������ǣ��ϸߵ��¶��ܹ��ӿ������Ӧ�����ʣ�Ҳʹ����Һ�в�����c��Mg2+����С�����Mg2+�ij����ʣ����¶ȹ���������Mg5��OH��2��CO3��4•4H2O��
��4����ϴ�Ӳ����У���������ϴ�Ӹɾ��IJ���Ϊ��ȡ���һ��ϴ����Һ�������м���HNO3�ữ��AgNO3��Һ�����ް�ɫ�������ɣ�����ϴ�Ӹɾ���
���� ��������±ˮ����Ҫ�ɷ�ΪMgCl2���л�ȡMgCO3•3H2O����±ˮ�м��������Ʋ�ͨ�������̼��̼��þ�����������ϴ�ӡ���ɡ���MgCO3•3H2O��
��1����������������MgCO3•3H2O�������ݵ���غ��Ԫ���غ���д��Ӧ�����ӷ���ʽ��
��2������ͼ1��֪���������̵�pH��ʱ��ı仯���ȱ�С������Ȼ����9-10֮�����ң�����Һ��ʼ��PHֵ��7-8���ң��ݴ��жϣ�
��3������ͼ2��֪���ϸߵ��¶��ܹ��ӿ������Ӧ�����ʣ�Ҳʹ����Һ�в�����c��Mg2+����С������ͼ3��֪��323Kʱ��Mg2+����Mg5��OH��2��CO3��4•4H2O������
��4����ϴ�Ӳ����У������ǴӺ��������ӵ���Һ�������ģ����Կ���ͨ�������������жϲ���ϴ���Ƿ�ɾ���
��� �⣺��1����������������MgCO3•3H2O������Ӧ�����ӷ���ʽΪMg2++CO2+2OH-+2H2O�TMgCO3•3H2O����
�ʴ�Ϊ��Mg2++CO2+2OH-+2H2O�TMgCO3•3H2O����
��2������ͼ1��֪���������̵�pH��ʱ��ı仯���ȱ�С������Ȼ����9-10֮�����ң�����Һ��ʼ��PHֵ��7-8���ң����Գ������̵IJ���Ϊ��±ˮ��ͨ��CO2�����ͣ�Ȼ��μ�NaOH��Һ��ͬʱ����ͨ��CO2����ѡc��
��3������ͼ2��֪���ϸߵ��¶��ܹ��ӿ������Ӧ�����ʣ�Ҳʹ����Һ�в�����c��Mg2+����С������ͼ3��֪��323Kʱ��Mg2+����Mg5��OH��2��CO3��4•4H2O����������ѡ����¶�Ϊ313K�������ǽϸߵ��¶��ܹ��ӿ������Ӧ�����ʣ�Ҳʹ����Һ�в�����c��Mg2+����С�����Mg2+�ij����ʣ����¶ȹ���������Mg5��OH��2��CO3��4•4H2O��
�ʴ�Ϊ��313K���ϸߵ��¶��ܹ��ӿ������Ӧ�����ʣ�Ҳʹ����Һ�в�����c��Mg2+����С�����Mg2+�ij����ʣ����¶ȹ���������Mg5��OH��2��CO3��4•4H2O��
��4����ϴ�Ӳ����У������ǴӺ��������ӵ���Һ�������ģ����Կ���ͨ�������������жϲ���ϴ���Ƿ�ɾ������Ա�������ϴ�Ӹɾ��IJ���Ϊȡ���һ��ϴ����Һ�������м���HNO3�ữ��AgNO3��Һ�����ް�ɫ�������ɣ�����ϴ�Ӹɾ���
�ʴ�Ϊ��ȡ���һ��ϴ����Һ�������м���HNO3�ữ��AgNO3��Һ�����ް�ɫ�������ɣ�����ϴ�Ӹɾ���
���� ���⿼���Ʊ�ʵ�鷽������ƣ�Ϊ�߿����㣬ȷ���ҵԭ��������ʵ���������Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬��Ŀ�ѶȲ���

| A�� | FeCl3����ǿ�����ԣ�ʹѪҺ�еĵ����ʱ���������ֹѪ | |
| B�� | FeCl3��ѪҺ������ѧ��Ӧ�����ɳ������ʶ�ֹѪ | |
| C�� | FeCl3ˮ������Fe��OH��3�����������ڻ��ƴ�ֹѪ | |
| D�� | FeCl3�ǵ���ʣ�ʹѪҺ�еĵ����ʺܿ��ɽ��ֹѪ |
| A�� | ƽ��������Ӧ�����ƶ� | B�� | A��ת�������� | ||
| C�� | m+n��p | D�� | C������������� |
| A�� | ��һ��Ԫ����ɵ�����һ���ǵ��� | |
| B�� | �κδ����ﶼ����һ��Ԫ����ɵ� | |
| C�� | ������Ԫ�صĻ�����һ���������� | |
| D�� | �в�ͬ��Ԫ����ɵĴ������ǻ����� |
| A�� | ʵ�����Ʊ�CO2�ķ�ӦΪ��CaCO3+2HCl�TCaCl2+H2O+CO2 | |
| B�� | ���Ȼ�ͭ��Һ�еμ���������Һ��Ag++Cl-�TAgCl | |
| C�� | ����������ϡ���ᷴӦ�����ӷ���ʽ��Ba2++OH-+H++SO42-�TBaSO4+H2O | |
| D�� | ϡ����ε�ͭƬ�ϣ�Cu+2H+�TCu2++H2 |
| A�� | c��H+��+c��Na+����c��CO32-��+c��HCO3-��+c��OH-�� | B�� | c��H+��+2c��H2CO3��+c��HCO3-��=c��OH-�� | ||
| C�� | c��Na+��=c��CO32-��+c��HCO3-��+c��H2CO3�� | D�� | c��Na+����c��CO32-����c��OH-����c��HCO3-����c��H+�� |

| A�� | ͼ1��ʾ���������Ƕȿ��ǣ�ʯī�Ƚ��ʯ�ȶ� | |
| B�� | ͼ2��ʾAl3+��OH-��Ӧʱ������Ũ�ȱ仯���ߣ�ͼ��a����Һ�д�������Al3+ | |
| C�� | ͼ3��ʾ��ͼ�е���Ӱ��������ĺ����ǣ�v��-v���� | |
| D�� | ͼ4��ʾ0.001 mol•L-1����ζ�0.001 mol•L-1NaOH��Һ�ĵζ����� |
| A�� | ϡ�����еμ�̼��������Һ | |
| B�� | ������Һ�еμӴ�����Һ | |
| C�� | ϡ������ʯ��ʯ��Ӧ | |
| D�� | �Ӳ�ľ������ȡ��̼������������ᷴӦ |
| A�� | þ����ǽ�������O2��Cl2�ȷ�Ӧ | B�� | þ�ڿ�����ȼ������Mg2O��Mg2O2 | ||
| C�� | ������þ��������ˮ�ļ� | D�� | ����þ�۵�ߣ����������²��� |