��Ŀ����
 �ס��ҡ���������������Ԫ�أ����м�Ԫ��ԭ�Ӻ���L����s�ܼ���p�ܼ����Ӹ�����ͬ����Ԫ��ԭ��3p�ܼ���ֻ��1�ԳɶԵ��ӣ����Ͷ�Ԫ��ԭ��N���϶�ֻ��1�����ӣ������б�Ԫ��ԭ�Ӹ��ڲ���ѳ���������Ԫ��ԭ�Ӵ����ĵ��ӳ�����2���ܼ��У���Ԫ�ص��ʳ���ʱΪ��̬��ԭ�����������Ų�ͼ��ͼ��ʾ���Իش��������⣮
�ס��ҡ���������������Ԫ�أ����м�Ԫ��ԭ�Ӻ���L����s�ܼ���p�ܼ����Ӹ�����ͬ����Ԫ��ԭ��3p�ܼ���ֻ��1�ԳɶԵ��ӣ����Ͷ�Ԫ��ԭ��N���϶�ֻ��1�����ӣ������б�Ԫ��ԭ�Ӹ��ڲ���ѳ���������Ԫ��ԭ�Ӵ����ĵ��ӳ�����2���ܼ��У���Ԫ�ص��ʳ���ʱΪ��̬��ԭ�����������Ų�ͼ��ͼ��ʾ���Իش��������⣮��1������
��2�������ʹ���ľ���������
��3����Ԫ��ԭ�����������Ų�ʽ��
��4�������Ȼ�����Һ���ҵ���̬�⻯�����Ӧ�����ӷ���ʽ�ǣ�
��5������ĵ��ʷ������Ϸ�Ӧ�Ļ�ѧ����ʽ�ǣ�
��6��д���Һ���ij�������̬������ͨ��������ʯ��ˮ�з����Ļ�ѧ����ʽ��
��7���������������Һ����ͨ�백��ֱ��������������������
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺
��������Ԫ��ԭ�Ӻ���L����s�ܼ���p�ܼ����Ӹ�����ͬ���ʼ�ΪC����Ԫ��ԭ��3p�ܼ���ֻ��1�ԳɶԵ��ӣ�����ΪS�����Ͷ�Ԫ��ԭ��N���϶�ֻ��1�����ӣ������б�Ԫ��ԭ�Ӹ��ڲ���ѳ���������Ԫ��ԭ�Ӵ����ĵ��ӳ�����2���ܼ��У��ʱ�ΪCu����ΪK����Ԫ��ԭ�����������Ų�ͼ��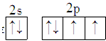 �����ĵ��ʳ���ʱΪ��̬��������O�����Ԫ�ض�Ӧ���ʡ�������������Լ���ĿҪ������⣮
�����ĵ��ʳ���ʱΪ��̬��������O�����Ԫ�ض�Ӧ���ʡ�������������Լ���ĿҪ������⣮
 �����ĵ��ʳ���ʱΪ��̬��������O�����Ԫ�ض�Ӧ���ʡ�������������Լ���ĿҪ������⣮
�����ĵ��ʳ���ʱΪ��̬��������O�����Ԫ�ض�Ӧ���ʡ�������������Լ���ĿҪ������⣮���
�⣺��Ԫ��ԭ�Ӻ���L����s�ܼ���p�ܼ����Ӹ�����ͬ���ʼ�ΪC����Ԫ��ԭ��3p�ܼ���ֻ��1�ԳɶԵ��ӣ�����ΪS�����Ͷ�Ԫ��ԭ��N���϶�ֻ��1�����ӣ������б�Ԫ��ԭ�Ӹ��ڲ���ѳ���������Ԫ��ԭ�Ӵ����ĵ��ӳ�����2���ܼ��У��ʱ�ΪCu����ΪK����Ԫ��ԭ�����������Ų�ͼ�� �����ĵ��ʳ���ʱΪ��̬��������O��
�����ĵ��ʳ���ʱΪ��̬��������O��
��1�������Ϸ�����֪��ΪC����ΪS����ΪCu����ΪK����ΪO���ʴ�Ϊ��C��S��Cu��K��O��
��2����ΪK�����ʹ���ľ��������ǽ������壬�ʴ�Ϊ���������壻
��3����Ϊͭ������29�����ӣ��۵����Ų�ʽΪ3d104s1���ʴ�Ϊ��3d104s1��
��4�������Ȼ�����Һ���Ȼ�ͭ���ҵ���̬�⻯��Ϊ���⣬���߷�����Ӧ������ͭ��������Ӧ�����ӷ���ʽ��Cu2++H2S�TCuS��+2H+��
�ʴ�Ϊ��Cu2++H2S�TCuS��+2H+��
��5��̼�������������Ϸ�Ӧ���ɶ�����̼����Ӧ�Ļ�ѧ����ʽ��C+O2
CO2���ʴ�Ϊ��C+O2
CO2��
��6���Һ���ij�������̬������ΪSO2�������ʯ��ˮ��Ӧ����������Ƴ���������ʽΪSO2+Ca��OH��2�TCaSO3��+H2O��
�ʴ�Ϊ��SO2+Ca��OH��2�TCaSO3��+H2O��
��7������ͭ�������İ�ˮ��Ӧ����������ͭ��������Ӧ���ӷ�Ӧ����ʽΪ��Cu2++2NH3?H2O�TCu��OH��2��+2NH4+������ͨ�백ˮ��������Ϸ�Ӧ�������ܽ⣬�γ�����ɫ����Һ��
�ʴ�Ϊ����ɫ��Һ��������ɫ�������氱���Ĺ�������ɫ�������ܽ⣬�γ�����ɫ����Һ��
 �����ĵ��ʳ���ʱΪ��̬��������O��
�����ĵ��ʳ���ʱΪ��̬��������O����1�������Ϸ�����֪��ΪC����ΪS����ΪCu����ΪK����ΪO���ʴ�Ϊ��C��S��Cu��K��O��
��2����ΪK�����ʹ���ľ��������ǽ������壬�ʴ�Ϊ���������壻
��3����Ϊͭ������29�����ӣ��۵����Ų�ʽΪ3d104s1���ʴ�Ϊ��3d104s1��
��4�������Ȼ�����Һ���Ȼ�ͭ���ҵ���̬�⻯��Ϊ���⣬���߷�����Ӧ������ͭ��������Ӧ�����ӷ���ʽ��Cu2++H2S�TCuS��+2H+��
�ʴ�Ϊ��Cu2++H2S�TCuS��+2H+��
��5��̼�������������Ϸ�Ӧ���ɶ�����̼����Ӧ�Ļ�ѧ����ʽ��C+O2
| ||
| ||
��6���Һ���ij�������̬������ΪSO2�������ʯ��ˮ��Ӧ����������Ƴ���������ʽΪSO2+Ca��OH��2�TCaSO3��+H2O��
�ʴ�Ϊ��SO2+Ca��OH��2�TCaSO3��+H2O��
��7������ͭ�������İ�ˮ��Ӧ����������ͭ��������Ӧ���ӷ�Ӧ����ʽΪ��Cu2++2NH3?H2O�TCu��OH��2��+2NH4+������ͨ�백ˮ��������Ϸ�Ӧ�������ܽ⣬�γ�����ɫ����Һ��
�ʴ�Ϊ����ɫ��Һ��������ɫ�������氱���Ĺ�������ɫ�������ܽ⣬�γ�����ɫ����Һ��
���������⿼��λ�á��ṹ�����ʵĹ�ϵ��Ӧ�ã�Ϊ��Ƶ���㣬���յ����Ų���Ԫ�ػ�����֪ʶ�ƶ�Ԫ��Ϊ���Ĺؼ������ط������ƶ������Ŀ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
����ͼʾʵ����ȷ���ǣ�������
A�� �ⶨNaOH��Һ��Ũ�� �ⶨNaOH��Һ��Ũ�� |
B�� ̼���������ȷֽ� ̼���������ȷֽ� |
C�� ����NH4Cl������Һ�Ʊ�NH4Cl���� ����NH4Cl������Һ�Ʊ�NH4Cl���� |
D�� �رշ�Һ©��������������Ͳ�������ɿ�������ָ�ԭ����֤��װ������������ �رշ�Һ©��������������Ͳ�������ɿ�������ָ�ԭ����֤��װ������������ |
������Һ���������ʵ�����ϵ��ȷ���ǣ�������
| A���������ʵ�����KHC2O4��H2C2O4����ˮ�����Һ��2c��K+��=c��HC2O4-��+c��H2C2O4�� |
| B��pH��ȵĢ�NH4Cl���ڣ�NH4��2SO4����NH4HSO4������Һ�У�c��NH4+����С����=�ڣ��� |
| C��0.1mol/LCH3COONa��Һ��0.15mol/LHCl�������ϣ�c��Cl-����c��H+����c��Na+����c��CH3COO-����c��OH-�� |
| D��0.1mol/L��KHA��Һ����pH=10��c��K+����c��A2-����c��HA-����c��OH-�� |

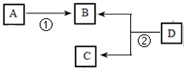 ��̬��A��һ�ֻ������л�����ԭ�ϣ�Ŀǰʯ���ѽ��ѳ�Ϊ����A����Ҫ������A�ڱ�״���µ��ܶ�Ϊ1.25g?L-1��D�ķ���ʽΪC4H8O2��D������������ˮ��õ�B��C��C����Է���������B��14�������л���֮���������ת����ϵ��
��̬��A��һ�ֻ������л�����ԭ�ϣ�Ŀǰʯ���ѽ��ѳ�Ϊ����A����Ҫ������A�ڱ�״���µ��ܶ�Ϊ1.25g?L-1��D�ķ���ʽΪC4H8O2��D������������ˮ��õ�B��C��C����Է���������B��14�������л���֮���������ת����ϵ��