��Ŀ����
9�� ��ͼװ���У�U����Ϊ��īˮ��a��b�Թ��ڷֱ�ʢ��ʳ��ˮ���Ȼ����Һ�������������飬����һ��ʱ�䣮
��ͼװ���У�U����Ϊ��īˮ��a��b�Թ��ڷֱ�ʢ��ʳ��ˮ���Ȼ����Һ�������������飬����һ��ʱ�䣮��1���������е�̼��������
��2�����Թ�����ͬ�ĵ缫��Ӧʽ�ǣ�Fe-2e-=Fe2+��
��3��a�Թ��з�����������ʴ�����ʴ���ͣ���ͬ�������ϵ缫��ӦΪ��O2+2H2O-4e-=4OH-��
b�Թ��з��������ⸯʴ�������ϵ缫��ӦΪ��2H++2e-=H2����
��4����īˮ�����ߵ�Һ��ı仯��U���ں�īˮ����ҵͣ�
���� �����к���̼���ʣ��ڵ���ʻ����£��������绯ѧ��ʴ������Һ�������������ⸯʴ������Һ����������������ʴ������Թ�����������Һ������������ʴ���ұ��Թ�����������Һ�������ⸯʴ���ݴ˽��
��� �⣺��1�������к���̼���ʣ���ԭ����У����ý�������������̼���������ʴ�Ϊ������
��2�����������绯ѧ��ʴʱ����������������ʧ���ӷ���������Ӧ��������ͬ�ĵ缫��ӦʽΪ��Fe-2e-=Fe2+��
�ʴ�Ϊ��Fe-2e-=Fe2+��
��3���ڵ���ʻ����£��������绯ѧ��ʴ������Թ�����������Һ������������ʴ������������ԭ��Ӧ��������ӦΪ��O2+2H2O-4e-=4OH-���ұ��Թ������Ȼ����Һ���Ȼ��ˮ�����Һ�����ԣ���Һ�������ⸯʴ��������ӦΪ��2H++2e-=H2����
�ʴ�Ϊ��������O2+2H2O-4e-=4OH-�����⣻2H++2e-=H2����
��4������Թ�����������Һ������������ʴ���ұ��Թ�����������Һ�������ⸯʴ����������Թ��������������С���ұ��Թ��������ѹǿ������U���ں�īˮ����ҵͣ�
�ʴ�Ϊ��U���ں�īˮ����ҵͣ�
���� ���⿼���˽����ĸ�ʴ���������ȷ��������������ʴ�����ⸯʴ�Ļ����ǽⱾ��ؼ����ѶȲ���

| A�� | ��״���£�11.2 L SO3��������ԭ����Ϊ1.5NA | |
| B�� | ��⾫��ͭʱ����������������6.4 g�����·��ת�Ƶ�����Ϊ0.2NA | |
| C�� | ���³�ѹ�£�4.0 g CH4�к��й��ۼ�����ĿΪNA | |
| D�� | �ں�Al3+����ΪNA��AlCl3��Һ�У�Cl-����Ϊ3NA |
| A�� | ��֪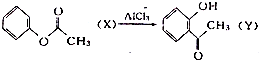 ��X��Y��Ϊͬ���칹�壬����FeCl3��Һ���� ��X��Y��Ϊͬ���칹�壬����FeCl3��Һ���� | |
| B�� |  �ܷ����ķ�Ӧ���ͣ��ӳɷ�Ӧ��ȡ����Ӧ����ȥ��Ӧ��ˮ�ⷴӦ �ܷ����ķ�Ӧ���ͣ��ӳɷ�Ӧ��ȡ����Ӧ����ȥ��Ӧ��ˮ�ⷴӦ | |
| C�� | 3-��-3-�һ������һ�ȴ�����3�� | |
| D�� | ��ͬ����������������ˮ�е��ܽ�ȱ������Ҵ��е��ܽ�ȴ� |
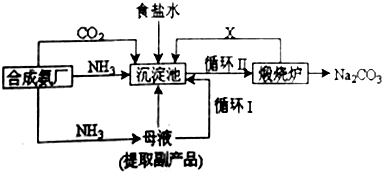
 ��ͼA��J�������������ˮ��Һ������B��D��G�ǵ��ʣ�B�ǵؿ��к�����ߵĽ���Ԫ�أ�G�����壬J�Ǵ��Բ��ϣ�������HΪ��ɫҺ�壮
��ͼA��J�������������ˮ��Һ������B��D��G�ǵ��ʣ�B�ǵؿ��к�����ߵĽ���Ԫ�أ�G�����壬J�Ǵ��Բ��ϣ�������HΪ��ɫҺ�壮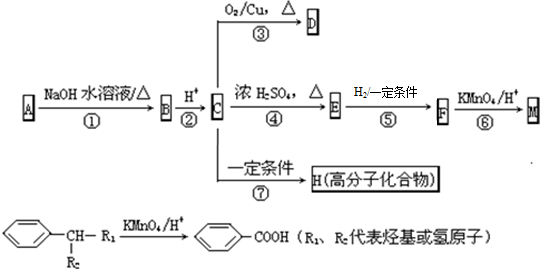
 ��
�� ��Ӧ�ߵĻ�ѧ����ʽΪ
��Ӧ�ߵĻ�ѧ����ʽΪ ��
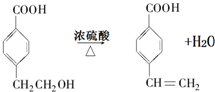
��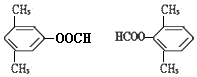 ��
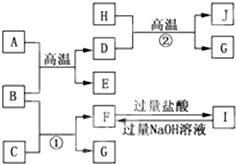
�� ��A��B��C��D��E��F������������A��E��������������Ԫ������Ҿ���10�����ӣ���������ͼ��ʾ��ת����ϵ��
��A��B��C��D��E��F������������A��E��������������Ԫ������Ҿ���10�����ӣ���������ͼ��ʾ��ת����ϵ��