��Ŀ����
5�������ʽṹ�����ʡ�Ԫ��Xλ�ڵ������ڣ����̬ԭ����4��δ�ɶԵ��ӣ�Yԭ�����������������ڲ����������3����Ԫ��Z��̬ԭ�ӵ�3p�������4�����ӣ�Wԭ�ӵ�2p�������3��δ�ɶԵ��ӣ�
��1��Y��W��Z���γɶ��ֻ����
��Ԫ��Y��Z�е縺�Խϴ����O����Ԫ�ط�����������
������ZY32-�Ŀռ乹��Ϊ�����Σ�������������������ԭ���ӻ���ʽsp3��
��ZY2�ռ乹��V�Σ�����ԭ���ӻ���ʽsp2�����ڼ��Է��ӣ�����ԡ��Ǽ��ԡ�����
��Ԫ��Y��W�е�һ�����ܽϴ����N����Ԫ�ط�����������
��2��Y���⻯�H2Y�����Ҵ��е��ܽ�ȴ���H2Z����ԭ����H2O�������Ҵ����Ӽ���γ��������H2S���ܣ�
��3����X2+����Һ��KCN����ˮ��Ӧ�ɵõ�������K3[X��CN��5��NH3��]��
�ٻ�̬X2+�ĵ����Ų�ʽ��1s22s22p63s23p63d6��
��1mol�����K3[X��CN��5��NH3��]�к��Ҽ�����ĿΪ14mol��
�ۻ���Xԭ�ӵļ۵����Ų�ͼ
 ��
����[X��CN��5��NH3��]3-�������д��ڵĻ�ѧ��������ac������ţ���
a����λ��b����� c�����Թ��ۼ�d���Ǽ��Թ��ۼ�e�����Ӽ���
���� Ԫ��Xλ�ڵ������ڣ����̬ԭ����4��δ�ɶԵ��ӣ���������Ų�ʽΪ1s22s22p63s23p63d64s2����XΪFe��Yԭ�����������������ڲ����������3����ԭ��ֻ����2�����Ӳ㣬����������Ϊ6����YΪOԪ�أ�Ԫ��Z��̬ԭ�ӵ�3p�������4�����ӣ���������Ų�ʽΪ1s22s22p63s23p4����ZΪSԪ�أ�Wԭ�ӵ�2p�������3��δ�ɶԵ��ӣ���������Ų�ʽΪ1s22s22p3����WΪNԪ�أ�
��� �⣺Ԫ��Xλ�ڵ������ڣ����̬ԭ����4��δ�ɶԵ��ӣ���������Ų�ʽΪ1s22s22p63s23p63d64s2����XΪFe��Yԭ�����������������ڲ����������3����ԭ��ֻ����2�����Ӳ㣬����������Ϊ6����YΪOԪ�أ�Ԫ��Z��̬ԭ�ӵ�3p�������4�����ӣ���������Ų�ʽΪ1s22s22p63s23p4����ZΪSԪ�أ�Wԭ�ӵ�2p�������3��δ�ɶԵ��ӣ���������Ų�ʽΪ1s22s22p3����WΪNԪ�أ�
��1����ͬ�������϶��µ縺�Լ�С���ʵ縺��O��S��
�ʴ�Ϊ��O��
������SO32-��Sԭ�ӹµ��Ӷ���=$\frac{6+2-2��3}{2}$=1���۲���Ӷ���=3+1=4���ռ�ṹΪ�����Σ�Sԭ�Ӳ�ȡsp3�ӻ���
�ʴ�Ϊ�������Σ�sp3��
��SO2������Sԭ�ӹµ��Ӷ���=$\frac{6-2��2}{2}$=1���۲���Ӷ���=2+1=3���ռ�ṹΪV�Σ�Sԭ�Ӳ�ȡsp2�ӻ�������������������IJ��غϣ�Ϊ���Է��ӣ�
�ʴ�Ϊ��V�Σ�sp2�����ԣ�
��NԪ��ԭ��2p���Ϊ�����ȶ�״̬�������ϵͣ���һ�����ܸ�����Ԫ�صģ�
�ʴ�Ϊ��N��
��2��H2O�������Ҵ����Ӽ���γ��������H2S���ܣ�H2O���Ҵ��е��ܽ�ȴ���H2S��
�ʴ�Ϊ��H2O�������Ҵ����Ӽ���γ��������H2S���ܣ�
��3���ٻ�̬Fe2+�ĵ����Ų�ʽ�ǣ�1s22s22p63s23p63d6��
�ʴ�Ϊ��1s22s22p63s23p63d6��
�ڰ�����������3���Ҽ���CN-�к���1���Ҽ����γ�6����λ����Ҳ���ڦҼ���1mol�����K3[Fe��CN��5��NH3��]�к�14mol�Ҽ���
�ʴ�Ϊ��14mol��
��Feԭ�ӵļ۵����Ų�ͼΪ�� ��
��
�ʴ�Ϊ�� ��
��
��[X��CN��5��NH3��]3-��������Fe2+��CN-��NH3�γ���λ����CN-��NH3�д��ڼ��Թ��ۼ���û����������Ӽ���
�ʴ�Ϊ��ac��
���� ���⿼���Ƕ����ʽṹ�����ʵĿ��飬�漰��������Ų����縺�ԡ������ܡ��ռ乹�����ӻ���ʽ�жϡ��������ѧ���ȣ�ע��ͬ���ڵ�һ�������쳣ԭ��

| A�� | K+��Na+��HCO${\;}_{3}^{-}$��Cl- | B�� | Fe3+��SCN-��Cl-��SO${\;}_{4}^{2-}$ | ||
| C�� | NH${\;}_{4}^{+}$��Fe2+��SO${\;}_{4}^{2-}$��NO${\;}_{3}^{-}$ | D�� | Mg2+��Fe2+��SO${\;}_{4}^{2-}$��Cl- |
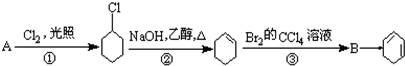
 ��B��������1��2-���廷���飮
��B��������1��2-���廷���飮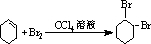 ��
��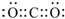 ��
��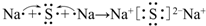 ��
��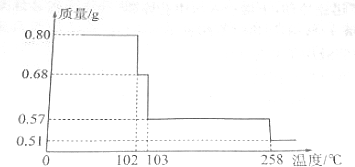
 ͼΪʵ����ijŨ�����Լ�ƿ�ϵı�ǩ���Ը����й����ݻش��������⣺
ͼΪʵ����ijŨ�����Լ�ƿ�ϵı�ǩ���Ը����й����ݻش��������⣺ �����������Ļ�ѧʽΪ��Cl2O7��
�����������Ļ�ѧʽΪ��Cl2O7��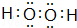 ��
��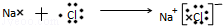 ��
��