��Ŀ����
(8��)����ʱ������ͬѧ�Ʊ�Fe(OH)3���壬����������£�
�������ձ��м���25 mL����ˮ���þƾ��Ƽ��������ڣ�Ȼ�����ձ�����εμ�6��FeCl3������Һ�������������Һ������ĺ��ɫ������Fe(OH)3���塣
�Իش��������⣺
��1��д���÷�Ӧ�Ļ�ѧ����ʽ________________________________________
��2�����֤������ͬѧ�Ƿ�ɹ��Ƶý��壿_________________________________________
��3��������ͬѧ�ڵμ�FeCl3������Һ��ͬʱ�ò��������Ͻ��裬���û�еõ����壬Ϊʲô�أ�_________________________________________________________________
��4������ͬѧ�Ƶõ�Fe(OH)3�����к����������ᣬΪ�õ��ϴ����Ľ��壬������������²�������������____ ____��
A������NaOH��Һ�����к�
B������AgNO3��Һ��Ӧ�����
C��װ���Ĥ���У��������ڣ�����ȫ����������ˮ�У���ÿ��һ��ʱ�䣬����һ������ˮ
��1��FeCl3 + 3H2O 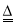 Fe(OH)3(����) + 3HCl
Fe(OH)3(����) + 3HCl
��2�����ý���Ķ����ЧӦ����һ���ɼ���ͨ�����÷�ɢϵ���������������ܿ��������ĵ�·����֤���ѳɹ��Ƶý��塣
��3�������ʹ��������۳�
��4��C��
��������
�����������1���÷�Ӧ������FeCl3������Һ��ȡFe(OH)3���壬����ʽΪ��FeCl3 + 3H2O 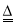 Fe(OH)3(����) + 3HCl ����2�����ý���Ķ����ЧӦ����һ���ɼ���ͨ�����÷�ɢϵ���������������ܿ��������ĵ�·����֤���ѳɹ��Ƶý��塣��2������ɼӿ콺�����˶����ʣ������˽���֮�����ײ���ᣬ ʹ���巢���۳�����3������NaOH��AgNO3�ȵ������Һ��ʹ���巢���۳������岻��ͨ����Ĥ��ˮ���Ӻ�������ͨ����Ĥ���ʿ����������ķ�������������ᴿ����C��ȷ��
Fe(OH)3(����) + 3HCl ����2�����ý���Ķ����ЧӦ����һ���ɼ���ͨ�����÷�ɢϵ���������������ܿ��������ĵ�·����֤���ѳɹ��Ƶý��塣��2������ɼӿ콺�����˶����ʣ������˽���֮�����ײ���ᣬ ʹ���巢���۳�����3������NaOH��AgNO3�ȵ������Һ��ʹ���巢���۳������岻��ͨ����Ĥ��ˮ���Ӻ�������ͨ����Ĥ���ʿ����������ķ�������������ᴿ����C��ȷ��
���㣺���齺����Ʊ������ʡ�

 ��У����ϵ�д�
��У����ϵ�д� H2(g)+Br2(g); ��H>0���ﵽƽ��ʱ��Ҫʹ���������ɫ����ɲ�ȡ�ķ���
H2(g)+Br2(g); ��H>0���ﵽƽ��ʱ��Ҫʹ���������ɫ����ɲ�ȡ�ķ��� pC(g) + qD(g) ��H<0������˵������ȷ����
pC(g) + qD(g) ��H<0������˵������ȷ����