��Ŀ����
���мס��ҡ�������ͬѧ�ֱ����Fe��OH��3������Ʊ�ʵ�飮
��ͬѧ��1mol?L-1�Ȼ�����Һ�м���������NaOH��Һ��
����ͬѧֱ�Ӽ��ȱ���FeCl3��Һ��
��ͬѧ��25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��
ֹͣ���ȣ�
����������ۺ�ʵ���Իش��������⣺
��1�����в�������ܳɹ���ͬѧ�� �����IJ������漰���Ļ�ѧ��Ӧ����ʽΪ ��
��2�������İ취ȷ֤��Fe��OH��3�������ɣ�������ǣ� �������ǣ� ��һ����Fe��OH��3�������ɣ�
��3����ͬѧ�������Ƶõ�Fe��OH��3�����������ʵ�飺
�ٽ���װ��U�ι��ڣ���ʯī���缫����ֱͨ���磬ͨ��һ��ʱ���������������ɫ�������� ��
����������������������������� ��
����������μ������NaHSO4��Һ�������� ��ԭ���� ��
��ͬѧ��1mol?L-1�Ȼ�����Һ�м���������NaOH��Һ��
����ͬѧֱ�Ӽ��ȱ���FeCl3��Һ��
��ͬѧ��25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��
ֹͣ���ȣ�
����������ۺ�ʵ���Իش��������⣺
��1�����в�������ܳɹ���ͬѧ��
��2�������İ취ȷ֤��Fe��OH��3�������ɣ�������ǣ�
��3����ͬѧ�������Ƶõ�Fe��OH��3�����������ʵ�飺
�ٽ���װ��U�ι��ڣ���ʯī���缫����ֱͨ���磬ͨ��һ��ʱ���������������ɫ��������
�����������������������������
����������μ������NaHSO4��Һ��������
���㣺�������Ҫ����
ר�⣺��Һ�ͽ���ר��
��������1��ʵ�����Ʊ����������������ڷ��ڵ�����ˮ�м��뱥���Ȼ�����Һ��
��2��������ж�������ʣ�������������ɢϵ�Ķ������ʣ�
��3���������Ӵ��е�ɣ����е�Ӿ�����ʣ��ܷ����۳���
��2��������ж�������ʣ�������������ɢϵ�Ķ������ʣ�
��3���������Ӵ��е�ɣ����е�Ӿ�����ʣ��ܷ����۳���
���
�⣺��1��ʵ�����Ʊ����������������ڷ��ڵ�����ˮ�м��뱥���Ȼ�����Һ������Һ��Ϊ���ɫʱ����ֹͣ���ȣ���Ӧ�Ļ�ѧ����ʽΪFeCl3+3H2O
Fe��OH��3�����壩+3HCl�������������������ɽ��壬�����õ�������
�ʴ�Ϊ������FeCl3+3H2O
Fe��OH��3�����壩+3HCl��
��2��������ж����ЧӦ�����ü��������ʱ������һ�������Ĺ�·��
�ʴ�Ϊ���ü�������䣻��һ�������Ĺ�·��
��3����Fe��OH��3���������磬ͨ��ʱ������ɵ������������ƶ�������������ɫ���
�ʴ�Ϊ��Fe��OH��3���������磻
�������м������������������SO42-ʹFe��OH��3���巢���۳������ɺ��ɫ������
�ʴ�Ϊ�����ɺ��ɫ�ij�����
��������������������μ������NaHSO4��Һ��NaHSO4�������SO42-ʹFe��OH��3���巢���۳���H+ʹFe��OH��3�����ܽ⣬��۲쵽�ȳ��ֺ��ɫ�������������ʧ��
�ʴ�Ϊ���ȳ��ֺ��ɫ�������������ʧ��NaHSO4�������SO42-ʹFe��OH��3���巢���۳���H+ʹFe��OH��3�����ܽ⣮
| ||
�ʴ�Ϊ������FeCl3+3H2O
| ||
��2��������ж����ЧӦ�����ü��������ʱ������һ�������Ĺ�·��
�ʴ�Ϊ���ü�������䣻��һ�������Ĺ�·��
��3����Fe��OH��3���������磬ͨ��ʱ������ɵ������������ƶ�������������ɫ���
�ʴ�Ϊ��Fe��OH��3���������磻
�������м������������������SO42-ʹFe��OH��3���巢���۳������ɺ��ɫ������
�ʴ�Ϊ�����ɺ��ɫ�ij�����
��������������������μ������NaHSO4��Һ��NaHSO4�������SO42-ʹFe��OH��3���巢���۳���H+ʹFe��OH��3�����ܽ⣬��۲쵽�ȳ��ֺ��ɫ�������������ʧ��
�ʴ�Ϊ���ȳ��ֺ��ɫ�������������ʧ��NaHSO4�������SO42-ʹFe��OH��3���巢���۳���H+ʹFe��OH��3�����ܽ⣮
���������⿼�齺����Ʊ������ʣ���Ŀ�ѶȲ����״���Ϊ������Ʊ���ע���Ʊ������������ص���ս���ľ۳������ʣ�

��ϰ��ϵ�д�
 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д�
�����Ŀ
���»�ѧҩƷ�����ʵ�������ȷ���ǣ�������
A�� ����Ũ���� ����Ũ���� |
B�� ��������������� ��������������� |
C�� ϡ��Ũ���� ϡ��Ũ���� |
D��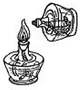 ��ȼ�ƾ��� ��ȼ�ƾ��� |
��һ���¶Ⱥ�ѹǿ�£�10L����A2��20L����B2��������20Lij����C��������C��ѧʽΪ��������
| A��AB2 |
| B��A2B |
| C��AB3 |
| D��AB |
�����������ʣ�������غ��Ȼ��ƹ��� ��39%�ľƾ���Һ �����ˮ��Һ���������ϸ���������ȷ���������ǣ�������
| A��������ȡ��Һ |
| B���ᾧ������ȡ |
| C���ᾧ��Һ���� |
| D���ᾧ�����Һ |
������������ȷ���ǣ�������
| A��H2SO4��Ħ��������98 |
| B����������O2��O3����������ԭ�Ӹ������ |
| C����������CO2��CO�������е�̼ԭ�Ӹ������ |
| D����98g H2SO4�ܽ���500mLˮ�У�������Һ��H2SO4�����ʵ���Ũ��Ϊ2mol/L |
���ӷ���ʽ�ܱ�ʾͬ���͵Ķ����Ӧ��һ�����������ӷ���ʽH++OH-=H2O��ʾ���ǣ�������
| A��2HCl+Ca��OH��2=CaCl2+2H2O |
| B��2NaOH+H2SO4=Na2SO4+2H2O |
| C��Ba��OH��2+2HNO3=Ba��NO3��2+2H2O |
| D��Cu��OH��2+2HCl=CuCl2+2H2O |
��NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A����״���£�1LҺ̬ˮ�к��е�H+��ĿΪ10-7NA |
| B����״���£�2.24LD2O�к��еĵ�����ΪNA |
| C��3.4gH2O2�к��еĹ��õ��Ӷ���Ϊ0.1NA |
| D��1mol̼ϩ����CH2���к��еĵ�����Ϊ8NA |
 2C(g)��D(g)������������A��B����4 mol����ǰ10����A��ƽ����Ӧ����Ϊ0��12 mol/(L��s)����10����ʱ��������B�����ʵ�����
2C(g)��D(g)������������A��B����4 mol����ǰ10����A��ƽ����Ӧ����Ϊ0��12 mol/(L��s)����10����ʱ��������B�����ʵ�����