��Ŀ����
�жϺ�����ǿ����һ��������ɣ���������ӽṹ�к����ǻ���ԭ����Խ�࣬�ú����������Խǿ������ʵ�����±���ʾ��
��1�������ᣨH3PO3���������ᣨH3AsO3���ķ���ʽ���ƣ������ǵ����Բ��ܴ�����������ǿ�ᣬ������������������������ԣ��ɴ˿��Ƴ����ǵĽṹʽ�ֱ�Ϊ�������� �������� ��
��2���ֱ�д����������������������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ������� ������� ��
| ������ | ���� | ���� | ������ | |
| ������ | Cl-OH | 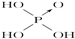 | 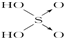 | 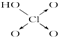 |
| ���ǻ� ��ԭ���� | 0 | 1 | 2 | 3 |
| ���� | ���� | ��ǿ�� | ǿ�� | ��ǿ�� |
��2���ֱ�д����������������������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ�������
���㣺��������������ʵ����ʵ����ϵ��Ӧ��,���ʵĽṹ������֮��Ĺ�ϵ
ר�⣺��ѧ���뾧��ṹ
��������1����������ӽṹ�к����ǻ���ԭ����Խ�࣬�ú����������Խǿ���ݴ�ȷ����Ľṹʽ��
��2����ͼӦ�����κ�ˮ���������к���2���ǻ������ڶ�Ԫ�ᣬ������������Ԫ�ᣮ
��2����ͼӦ�����κ�ˮ���������к���2���ǻ������ڶ�Ԫ�ᣬ������������Ԫ�ᣮ
���
�⣺��1����������ӽṹ�к����ǻ���ԭ����Խ�࣬�ú����������Խǿ������������ǿ�ᣬ������������������������ԣ������������������Ľṹʽ�ֱ�Ϊ�� ��
�� ��
��
�ʴ�Ϊ�� ��
�� ��
��
��2����ͼӦ�����κ�ˮ���������к���2���ǻ������ڶ�Ԫ�ᣬ������������Ԫ�ᣬ�������������ֱ���������Ƶķ�Ӧ����ʽΪ��H3PO3+2NaOH=Na2HPO3+2H2O��H3AsO3+3NaOH=Na3AsO3+3H2O��
�ʴ�Ϊ��H3PO3+2NaOH=Na2HPO3+2H2O��H3AsO3+3NaOH=Na3AsO3+3H2O��
 ��
�� ��
���ʴ�Ϊ��
 ��
�� ��
����2����ͼӦ�����κ�ˮ���������к���2���ǻ������ڶ�Ԫ�ᣬ������������Ԫ�ᣬ�������������ֱ���������Ƶķ�Ӧ����ʽΪ��H3PO3+2NaOH=Na2HPO3+2H2O��H3AsO3+3NaOH=Na3AsO3+3H2O��
�ʴ�Ϊ��H3PO3+2NaOH=Na2HPO3+2H2O��H3AsO3+3NaOH=Na3AsO3+3H2O��
���������⿼����������ʣ���ȷ������ǿ�����жϷ������ɽ���ѶȲ���

��ϰ��ϵ�д�
 �ƸԴ��ž�ϵ�д�
�ƸԴ��ž�ϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A�������½�pH=2�Ĵ����pH=12��NaOH��Һ�������ϣ�������Һ�Լ��� |
| B����֪ͬ��ʱ�����ĵ��볣������������ĵ��볣��������ͬ�¶���ͬŨ�ȵ�NaF��Һ��NaNO2��Һ�ļ��ԣ�NaF��Һ��ǿ |
| C���������������Ͱ�ˮ��ϣ���ַ�Ӧ��������Һ�����ԣ�������Һ�����ʵ���Ũ�ȹ�ϵΪ��c��HCl����c��NH3?H2O�� |
| D�������£�pH��Ϊ5�Ĵ������������Һ����ˮ�������������Ũ�Ⱦ�Ϊ1��10-9 mol?L-1 |
��������ķ�Ӧ 14CuSO4+5FeS2+12H2O�T7Cu2S+5FeSO4+12H2SO4������˵����ȷ���ǣ�������
| A��������ֻ��CuSO4 |
| B��Cu2S �������������ǻ�ԭ�� |
| C�������������뱻��ԭ�����������Ϊ3��7 |
| D��14mol CuSO4������5mol FeS2 |
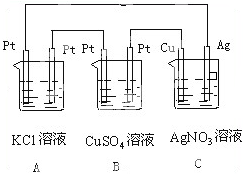 ��ͼ��ʾ������Ӧһ��ʱ�䣬5minʱͭ�缫��������1.08g���Իش�
��ͼ��ʾ������Ӧһ��ʱ�䣬5minʱͭ�缫��������1.08g���Իش�