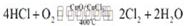��Ŀ����
(12��)��Cl2����ijЩ�����л���ʱ�����������HC1�����÷�ӦA����ʵ���ȵ�ѭ�����á�
��ӦA:
��1����֪:i ��ӦA�У� 4mol HCI���������ų�115.6kJ��������
ii
��H2O�ĵ���ʽ��_______________.
�ڷ�ӦA���Ȼ�ѧ����ʽ��_______________��
�۶Ͽ�1 mol H��O ����Ͽ� 1 mol H��Cl �������������ԼΪ__________kJ��H2O��
H��O ����HCl��H��Cl�����ǿ����������_______________��
��2�����ڷ�ӦA����ͼ��4��Ͷ�ϱ�[n��HCl����n��O2�����ֱ�Ϊ1��1��2��1��4��1��6��1]�£���Ӧ�¶ȶ�HClƽ��ת����Ӱ������ߡ�
������b��Ӧ��Ͷ�ϱ���______________.
�ڵ�����b��c��d��Ӧ��Ͷ�ϱȴﵽ��ͬ��HClƽ��ת����ʱ����Ӧ�ķ�Ӧ�¶���Ͷ
�ϱȵĹ�ϵ��_________________.
��Ͷ�ϱ�Ϊ2:1���¶�Ϊ400��ʱ��ƽ��������Cl2�����ʵ���������_______________.
����1����4HCl(g)+O2(g)
2Cl2(g)+2H2O(g) ��H= - 115.6 kJ/mol��32��ǿ����2��4:1��Ͷ�ϱ�ԽСʱ��Ӧ���¶�Խ�ͣ�30.8%��
����:����1������ˮ���ӵĽṹ�������ʽΪ������ӦA���Ȼ�ѧ����ʽΪ��4HCl(g)+O2(g)
2Cl2(g)+2H2O(g) ��H= - 115.6 kJ/mol�����ڷ�Ӧ�У���Ҫ����4mol��H��Cl�������Ͽ�1mol��O=O�������γ�2mol��Cl-Cl�������γ�4mol��H-O����������ͼֽ���ݣ��Ͽ�1mol��O=O�������γ�2mol��Cl-Cl������������Ϊ12kJ/mol������Ӧ����ЧӦΪ��-115.6 kJ/mol���ʴ˶���4mol��H��Cl�������γ�4mol��H-O������������Ϊ��-127.6 kJ/mol���ʶϿ�1 mol H��O ����Ͽ� 1 mol H��Cl �������������ԼΪ32kJ��H2O��H��O ����HCl��H��Cl��ǿ����2�����ݷ�Ӧ����ʽ��4HCl+O2
2Cl2+2H2O��������Ͷ�ϱ�Խ�ߣ���HCl��ת����Խ�ߣ��ʴ�����b��Ӧ��Ͷ�ϱ�Ϊ4:1�����ڸ÷�Ӧ������ȣ����¶�Խ�ߣ�HCl��ת����Խ�ͣ���Ͷ�ϱ�ԽСʱ�¶�Խ�ߣ���Ͷ�ϱ�Ϊ2:1���¶�Ϊ400��ʱ��HCl��ת����ԼΪ80%����ʱΪ��
4HCl+O22Cl2+2H2O��
��ʼ 2 1 0 0
�仯 1.6 0.4 0.8 0.8
ƽ�� 0.4 0.6 0.8 0.8����ƽ�����������������ʵ�������Ϊ0.8/2.6=30.8%��

 �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д�