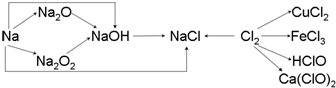
��Ŀ����
2�����ڱ���ǰ�����ڵ�Ԫ��A��B��C��D��E�����ǵĺ˵����������������A��B��C��ͬһ���ڵķǽ���Ԫ�أ�������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��AC2Ϊ�Ǽ��Է��ӣ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ�E��ԭ������Ϊ24��ECl3����B��C���⻯���γ�����λ��������������������ʵ���֮��Ϊ2��1������������λ����磮���������������ش��������⣺������ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ����1��B���⻯�������ԭ�ӵļ۲���ӶԻ��⣨VSEPR��ģ���������壬����ԭ���ӻ���ʽΪsp3�����ӵĿռ乹���������Σ�
��2��A��ͬ���������ж��֣�����һ���ʵ����������ܵ��磬��1molAԭ�ӵĸ������ЦҼ�����ĿΪ1.5NA��
��3����B��C��ɵ�һ�ֻ�������AC2��Ϊ�ȵ����壬�仯ѧʽΪN2O��
��4��E�ĺ���۵��Ӳ��Ų�ʽ��3d54s1��ECl3�γɵ������Ļ�ѧʽΪ[Cr��NH3��4��H2O��2]Cl3��
��5��B������������Ӧ��ˮ�����ϡ��Һ��D�ĵ��ʷ�Ӧʱ��B����ԭ����ͼۣ��÷�Ӧ�Ļ�ѧ����ʽ��4Mg+9HNO3=4Mg��NO3��2+NH3��+3H2O��
���� ���ڱ���ǰ�����ڵ�Ԫ��A��B��C��D��E�����ǵĺ˵����������������A��B��C��ͬһ���ڵķǽ���Ԫ�أ�
������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����D��C����һ���ڣ���D�ǵ�IIA��Ԫ�أ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ����γ�������⻯���а�����ˮ��HF��AC2Ϊ�Ǽ��Է��ӣ�AΪ��IVA�壬CΪ��VIA�壬��A��C��B��N��C��OԪ�أ�D��MgԪ�أ�
E��ԭ������Ϊ24����E��CrԪ�أ�CrCl3����N��O���⻯���γ�����λ��������������������ʵ���֮��Ϊ2��1������������λ����磬�������ﻯѧʽΪ[Cr��NH3��4��H2O��2]Cl3���ٽ����Ŀ�������
��� �⣺���ڱ���ǰ�����ڵ�Ԫ��A��B��C��D��E�����ǵĺ˵����������������A��B��C��ͬһ���ڵķǽ���Ԫ�أ�
������DC�ľ���Ϊ���Ӿ��壬D�Ķ�����������C�������Ӿ�����ͬ�ĵ��Ӳ�ṹ����D��C����һ���ڣ���D�ǵ�IIA��Ԫ�أ�B��C���⻯��ķе������ͬ����������Ԫ���⻯��ķе�ߣ����γ�������⻯���а�����ˮ��HF��AC2Ϊ�Ǽ��Է��ӣ�AΪ��IVA�壬CΪ��VIA�壬��A��C��B��N��C��OԪ�أ�D��MgԪ�أ�
E��ԭ������Ϊ24����E��CrԪ�أ�CrCl3����N��O���⻯���γ�����λ��������������������ʵ���֮��Ϊ2��1������������λ����磬�������ﻯѧʽΪ[Cr��NH3��4��H2O��2]Cl3��
��1��N���⻯����NH3��������������ԭ��Nԭ�Ӽ۲���ӶԸ�����4�Һ���һ���µ��Ӷԣ������������е�����ԭ�ӵļ۲���ӶԻ��⣨VSEPR��ģ�����������Σ�����ԭ���ӻ���ʽΪsp3�����ӵĿռ乹���������Σ��ʴ�Ϊ�������壻sp3��������
��2��A��ͬ���������ж��֣�����һ���ʵ����������ܵ��磬Ϊʯī��ʯī��ÿ��Cԭ���γ�1.5���Ҽ�������1molAԭ�ӵĸ������ЦҼ�����ĿΪ1.5NA��
�ʴ�Ϊ��1.5NA��
��3����B��C��ɵ�һ�ֻ�������CO2��Ϊ�ȵ����壬�ȵ������ԭ�Ӹ�������Ҽ۵�������ȣ������仯ѧʽΪN2O���ʴ�Ϊ��N2O��
��4��E��CrԪ�أ���3d��4s����Ϊ��۵��ӣ�����E�ĺ���۵��Ӳ��Ų�ʽ��3d54s1��ͨ�����Ϸ���֪��ECl3�γɵ������Ļ�ѧʽΪ[Cr��NH3��4��H2O��2]Cl3���ʴ�Ϊ��3d54s1��[Cr��NH3��4��H2O��2]Cl3��
��5��N������������Ӧ��ˮ�����ϡ��Һ��ϡ���ᣬ��D�ĵ��ʷ�Ӧʱ��B����ԭ����ͼۣ�Ϊ�仹ԭ�����ǰ�����D����þ���ӣ��÷�Ӧ�Ļ�ѧ����ʽ��4Mg+9HNO3=4Mg��NO3��2+NH3��+3H2O��
�ʴ�Ϊ��4Mg+9HNO3=4Mg��NO3��2+NH3��+3H2O��
���� ���⿼��λ�ýṹ�������ϵ��Ӧ�ã��漰ԭ���ӻ���ʽ�жϡ����ռ乹���жϡ��ȵ����塢ԭ�Ӻ�������Ų���������ԭ��Ӧ��֪ʶ�㣬�ۺ��Խ�ǿ��ע��۲���ӶԻ������۵�������ã��״�����Crԭ�Ӽ۵����Ų�ʽ����д����Ŀ�ѶȲ���

| A�� | ľ̿Ͷ�뵽�ȵ�Ũ������ | B�� | ľ̿Ͷ�뵽�ȵ�Ũ������ | ||
| C�� | ͭƬͶ�뵽���Ũ������ | D�� | ͭƬͶ�뵽���Ũ������ |
| A�� | ���������Ʊ�����ú���� | |
| B�� | ��������������Һʱ��Ҫ�����м���������������� | |
| C�� | Ũ�����ô�������ϸ�ڡ���ɫ�Լ�ƿʢ�ţ��������������� | |
| D�� | �Լ�ƿ�е�Һ�����ˮ��棬��ֹ��ӷ� |
| A�� | ���ڳ�ʪ�Ļ��������⣺3Fe+4H2O=Fe3O4+4H2 | |
| B�� | ʢ���ռ���Һ���Լ�ƿ�����ò�������SiO2+2NaOH=Na2SiO3+H2O | |
| C�� | ������ͭ���ڹ�����ˮ��Cu��OH��2+4NH3=[Cu��NH3��4]��OH��2 | |
| D�� | ��ú�м���ʯ��ʯ�ɼ���úȼ��ʱSO2���ŷţ�2CaCO3+O2+2SO2$\frac{\underline{\;\;��\;\;}}{\;}$2CaSO4+2CO2 |
| A�� | �٢ۢ� | B�� | �٢ڢۢ� | C�� | �ڢۢ� | D�� | �٢ۢܢ� |
| A�� | ����A��B�������ʵķ����ж���һ����ͬ��ʽ������45��ԭ���� | |
| B�� | A��ʽ����B��14 | |
| C�� | B��ʽ����A��16 | |
| D�� | ������һ������˫���Ȳ����ͼ� |