��Ŀ����
13��������ԭ��Ӧ��������������ʹ�÷dz��㷺��I��NaNO2��ۺ�ʳ�����ƣ�������ζ������ʹ����ʳ�ж�����֪NaNO2�ܷ������·�Ӧ��
2NaNO2+4HI�T2NO��+I2+2NaI+2H2O��
��1��������Ӧ����������NaNO2��
��2������������Ӧ������NaNO2��NaCl����ѡ�õ������У��ٵ⻯�ص�����ֽ �ڵ��� �۰� ��ʳ�ף�����ΪӦѡ�õ������Т٢ܣ�����ţ���
��3��ij����Һ�У�����2%��5%��NaNO2��ֱ���ŷŻ������Ⱦ�������Լ���ʹNaNO2ת��Ϊ�����������Ⱦ��N2����B��
A��NaCl B��NH4Cl C��HNO3 D��ŨH2SO4
II��������������˫��ˮ��ҽ������������ɱ��������������ϴ�˿ڣ��������з�Ӧ�ش����⣺
A��Na2O2+2HCl�T2NaCl+H2O2
B��Ag2O+H2O2�T2Ag+O2��+H2O
C.2H2O2�T2H2O+O2��
D.3H2O2+Cr2��SO4��3+10KOH�T2K2CrO4+3K2SO4+8H2O
��1��������Ӧ�У�H2O2�����������Եķ�Ӧ�ǣ�����ţ���ͬ��D��H2O2�����������������ֻ�ԭ�Եķ�Ӧ��C��
��2��������Ӧ˵��H2O2��Ag2O��K2CrO4����������ǿ������˳����Ag2O��H2O2��K2CrO4
��3��ij���Է�Ӧ��ϵ�з�����һ��������ԭ�����ӷ�Ӧ����Ӧ��������ﹲ��������O2��MnO4-��H2O��Mn2+��H2O2��H+����֪�÷�Ӧ��H2O2ֻ���������¹��̣�H2O2��O2��
�ٽ��������ͻ�ԭ���Ļ�ѧʽ������ƽ���ϵ���������з����С�+��
�����������Ӧ����6.72L����״�����������ɣ�ת�Ƶĵ���Ϊ0.6mol��
��4��H2O2��ʱ����Ϊ��ҵ��Һ���������С���ɫ�������������ƣ��������ɿ�ҵ��Һ�е��軯���KCN���������·�Ӧ��KCN+H2O2+H2O�TA+NH3������ָ��������A�Ļ�ѧʽΪKHCO3��
���� I����1��������ԭ��Ӧ�У����ϼ۽���Ԫ�����ڵķ�Ӧ������������
��2�����ݷ�Ӧʱ�г��������塢��ɫ�仯����������ѡ���Լ���
��3��ѡ���Լ�ʱҪ���ǣ�����ʹNaNO2ת����������Ⱦ��
II����1��˫��ˮ����������˵��˫��ˮ��Ԫ�ػ��ϼ����ߣ����ֻ�ԭ��˵��˫��ˮ��Ԫ�ػ��ϼ۽��ͣ�H2O2�����������ᣬ�����ԣ����Ƕ�Ԫ���
��2���������������ԣ���������������ԣ���ԭ���������ԣ�
��3���ٸ���������ԭ��Ӧ���������ͻ�ԭ�����ص��������Ѱ�ҷ�Ӧ��������Ȼ���ϻ��ϼ����������ƽ��
�ڸ����������ķ�Ӧ���������ϼ۵ı仯����������õ�ת�Ƶ��ӵ���Ŀ��
��4������ԭ���غ���ȷ�����ʵĻ�ѧʽ��˫��ˮ���������Ļ�ԭ����Ϊˮ������Ⱦ���
��� �⣺I����1��2NaNO2+4HI�T2NO+I2+2NaI+2H2O����Ԫ�صĻ��ϼ۽��ͣ�����NaNO2����������
�ʴ�Ϊ��NaNO2��
��2�����ۡ�����NaNO2������Ӧ����������������KI��NaNO2��Ӧ����I2���������������²��ܷ�Ӧ��ʳ���������������ӣ��⻯����ʳ�ױ��������ܼ�ˮ�е���������ӣ��������Ƶ������������������ӣ�������Ӧ���ɵⵥ�ʣ�I2ʹʪ��ĵ��۵⻯����ֽ������
�ʴ�Ϊ���٢ܣ�
��3��NaNO2��N2�DZ���ԭ������ӻ�ԭ����NԪ������������O��ClԪ�أ���ֻ�ܼ�NH4Cl����ԭ��������ʽΪNaNO2+NH4Cl�TNaCl+N2��+2H2O�����������е��õ�3����ָ�����еĵ����Ȼ���е�ʧȥ3����ָ�����еĵ���
�ʴ�Ϊ��B��
II����1��D��˫��ˮ����Ԫ�ػ��ϼ۽��ͣ�˵��˫��ˮ���������������������ԣ�C��˫��ˮ����Ԫ�ػ��ϼ����ߣ����ֻ�ԭ�ԣ�
�ʴ�Ϊ��D��C��
��2����ӦAg2O+H2O2=2Ag+O2��+H2O�У�����������������˫��ˮ����ԭ������������Ag2O��H2O2����Ӧ3H2O2+Cr2��SO4��3+10KOH=2K2CrO4+3K2SO4+8H2O�У�˫��ˮ��������������������������������H2O2��K2CrO4����������˳��Ϊ��Ag2O��H2O2��K2CrO4��
�ʴ�Ϊ��Ag2O��H2O2��K2CrO4��
��3���ٸ÷�Ӧ�����У���������������Ժ�˫��ˮ�Ļ�ԭ�Ե��¶��䷢��������ԭ��Ӧ�����������������Ԫ�ش�+7��Ϊ+2�ۣ����ϼ۽���5�ۣ�˫��ˮ����Ԫ�ػ��ϼ۴�-1�۱�Ϊ0�ۣ����ϼ���������2�ۣ������������ӵ�ϵ��Ϊ2��˫��ˮ��ϵ��Ϊ5��Ȼ�����������غ���ƽ�ɵø÷�Ӧ�����ӷ���ʽΪ��2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2����
�ʴ�Ϊ��2MnO4-+5H2O2+6H+=2Mn2++8H2O+5O2����
��������Ӧ������5mol����ת�Ƶ���10mol������Ӧ����6.72L����״������0.3mol��������ʱ����ת�Ƶĵ���Ϊ��10mol��$\frac{0.3mol}{5}$=0.6mol��
�ʴ�Ϊ��0.6��
��4����ӦKCN+H2O2+H2O�TA+NH3���У�����ԭ���غ��֪������A�Ļ�ѧʽΪKHCO3��
�ʴ�Ϊ��KHCO3��
���� ���⿼��������ԭ��Ӧ����ƽ�����㡢��������ȣ��ѶȲ���ע��Ի���֪ʶ���������գ�

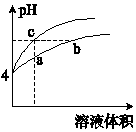 ij�¶��£������pH����ͬ��������Ȼ����Һ��ˮϡ��ʱ��pH�仯������ͼ��ʾ�������ж���ȷ���ǣ�������
ij�¶��£������pH����ͬ��������Ȼ����Һ��ˮϡ��ʱ��pH�仯������ͼ��ʾ�������ж���ȷ���ǣ�������| A�� | a��c������Һ�ĵ���������ͬ | |
| B�� | a��b��c������Һ��ˮ�ĵ���̶ȣ�a��b��c | |
| C�� | b����Һ�У�c��H+��+c��NH3•H2O��=c��OH-�� | |
| D�� | ��NaOH��Һ�͵����b��c����Һ��Ӧ������NaOH��Һ�����Vb=Vc |
| A�� | ��NaAlO2��Һ��ͨ������CO2��2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32- | |
| B�� | �Ȼ����Һ�����ʯ��ˮ��Ӧ��NH4++OH-?NH3��+H2O | |
| C�� | �⻯������������ˮ��Ӧ��2I-+Br2�TI2+2Br- | |
| D�� | ��������ˮ�����ӷ���ʽ��Cl2+H2O?2H++Cl-+ClO- |
| A�� | ȡһ�������壬�ܽ⣬����Һ�еμ�����AgNO3��Һ���۲쵽�а�ɫ�������ɣ�˵��ԭ������һ������NaCl | |
| B�� | ȡһ�������壬�ܽ⣬����Һ�м�������CaO��ĩ����ַ�Ӧ��۲쵽�а�ɫ�������ɣ�˵��ԭ������һ������Na2CO3 | |
| C�� | ȡһ�������壬�ܽ⣬����Һ��ͨ��������CO2���۲쵽�о���������˵��ԭ������һ������Na2CO3 | |
| D�� | ��ȡ3.80g���壬���������أ�����������0.620g��������ϡ�����ܽ�������壬��ַ�Ӧ���ռ���0.880g���壬˵��ԭ�����н�����Na2CO3��NaHCO3 |
 ��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺
��50mL0.50mol/L������50mL0.55mol/LNaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��ش��������⣺