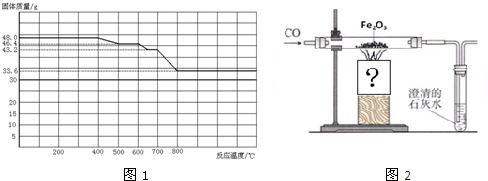
��Ŀ����
������һ���ḻ���ʱ��⣬ͨ����ˮ���ۺ����ÿɻ���������ʹ�����ʹ�ã�
��1����ˮ���εĿ������ã�
�ٺ�ˮ����Ŀǰ�����Ϊ�������������ѡ��Զ�뽭���뺣�ڣ�������꣬��ϫ��������ƽ̹�տ��ĺ�̲�����������Ϊ��ˮ�ء������غ� �أ�
��Ŀǰ��ҵ�ϲ��ñȽ��Ƚ������ӽ���Ĥ���۷������ȼҵ�������ڵ����������ӽ���Ĥֻ����������ͨ������ֹ�����Ӻ�����ͨ������˵���ȼ������������ӽ���Ĥ�����ã� ��дһ�㼴�ɣ���
��2�����������ǽ��귢չ���һ�ֽϺõĺ�ˮ������������ԭ����ͼ��ʾ�����о���ѡ���Ե������ӽ���Ĥ�������ӽ���Ĥ������У���ش���������⣺
�ٺ�ˮ����ֱ��ͨ�뵽�������У������� ��
��A���ų����� �����ˮ����Ũˮ������

��3���ÿ�±����Na+��K+��Mg2+��Cl-��Br-�����ӣ�����ȡ�壬�������������£�
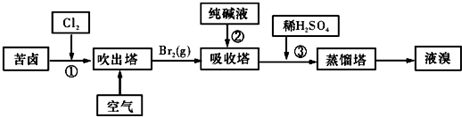
�����������е���Һ��BrO3-�����������з�Ӧ�����ӷ���ʽΪ�� ��
��ͨ�����Ȼ��ѻ�ú�Br2����Һ��Ϊ�λ��辭�����������ա��ữ���»�ú�Br2����Һ�� ��
������������ͨ��ˮ�������ȣ������¶���90�����ҽ��������ԭ���� ��
��1����ˮ���εĿ������ã�
�ٺ�ˮ����Ŀǰ�����Ϊ�������������ѡ��Զ�뽭���뺣�ڣ�������꣬��ϫ��������ƽ̹�տ��ĺ�̲�����������Ϊ��ˮ�ء������غ�
��Ŀǰ��ҵ�ϲ��ñȽ��Ƚ������ӽ���Ĥ���۷������ȼҵ�������ڵ����������ӽ���Ĥֻ����������ͨ������ֹ�����Ӻ�����ͨ������˵���ȼ������������ӽ���Ĥ�����ã�
��2�����������ǽ��귢չ���һ�ֽϺõĺ�ˮ������������ԭ����ͼ��ʾ�����о���ѡ���Ե������ӽ���Ĥ�������ӽ���Ĥ������У���ش���������⣺
�ٺ�ˮ����ֱ��ͨ�뵽�������У�������
��A���ų�����

��3���ÿ�±����Na+��K+��Mg2+��Cl-��Br-�����ӣ�����ȡ�壬�������������£�

�����������е���Һ��BrO3-�����������з�Ӧ�����ӷ���ʽΪ��
��ͨ�����Ȼ��ѻ�ú�Br2����Һ��Ϊ�λ��辭�����������ա��ữ���»�ú�Br2����Һ��
������������ͨ��ˮ�������ȣ������¶���90�����ҽ��������ԭ����
���㣺��ˮ��Դ�����ۺ�����
ר�⣺
��������1���ٺ�ˮɹ��ͨ���������ᾧ��ȡʳ�Σ�
�������ӽ���Ĥֻ��������ͨ���������Ӻ����岻��ͨ����
��2���ٺ�ˮ�к��е�Mg2+��Ca2+�������������������ӷ�Ӧ���ɳ�����
���ڵ�������������ˮʾ��ͼ��������������������������������������Ũ�ȴ��ΪŨˮ������Ũ��С��Ϊ��ˮ��
��3������������̼����������嵥�ʷ�Ӧ���������ӡ���������ӺͶ�����̼���壬�ݴ�д����Ӧ�����ӷ���ʽ��
���ڢ����嵥��Ũ�Ƚ�С��ͨ�����������ա��ữ���»�ú�Br2����Һ�����Ը����壬���ͳɱ���
���¶ȹ��ᵼ��ˮ���������������¶ȹ�������������������
�������ӽ���Ĥֻ��������ͨ���������Ӻ����岻��ͨ����
��2���ٺ�ˮ�к��е�Mg2+��Ca2+�������������������ӷ�Ӧ���ɳ�����
���ڵ�������������ˮʾ��ͼ��������������������������������������Ũ�ȴ��ΪŨˮ������Ũ��С��Ϊ��ˮ��
��3������������̼����������嵥�ʷ�Ӧ���������ӡ���������ӺͶ�����̼���壬�ݴ�д����Ӧ�����ӷ���ʽ��
���ڢ����嵥��Ũ�Ƚ�С��ͨ�����������ա��ữ���»�ú�Br2����Һ�����Ը����壬���ͳɱ���
���¶ȹ��ᵼ��ˮ���������������¶ȹ�������������������
���
�⣺��1���ٺ�ˮɹ����Ҫͨ���������ᾧ���̣����Ի���Ҫ�ᾧ�أ�
�ʴ�Ϊ���ᾧ��
�������ӽ���Ĥֻ��������ͨ���������Ӻ����岻��ͨ������ʯī������ⱥ���Ȼ���ʱ�������������ӷŵ�������������������ͨ�������ӽ���Ĥ�����������������������������������ϲ�����ը����������������Һ��Ӧ�����Ȼ��ơ��������ƶ�������ȡ���������Ʋ�����
�ʴ�Ϊ����ֹH2��Cl2������Ӧ����������ը����ֹCl2�����ɵ�NaOH��Һ��Ӧ��ʹ�ռ��Ʒ�����ȣ�
��2���ٺ�ˮ�к��к�ˮ�к��϶�Mg2+��Ca2+�������ӣ����ʱ���������������������ӣ��������������Ӻ�þ���ӷ�Ӧ����Mg��OH��2��Ca��OH��2�ȳ����Ӷ����������ӽ���Ĥ���Ӷ���������ͨ�������µ�������ʧ�ܣ�
�ʴ�Ϊ����ˮ�к��϶�Mg2+��Ca2+�������ӣ����ʱ�����Mg��OH��2��Ca��OH��2�ȳ����Ӷ����������ӽ���Ĥ��
���ڵ�������������ˮʾ��ͼ������������������������������������������Ũ�ȱ�С����������Ũ�Ȳ�������Ũ�ȴ��ΪŨˮ������Ũˮ��B���ų�����ˮ��A���ų���
�ʴ�Ϊ����ˮ��
��3���ٴ�����̼���ƣ����巴Ӧ��BrO3-���ɣ���Ӧ�����ӷ���ʽΪ��3CO32-+3Br2=5Br-+BrO3-+3CO2����
�ʴ�Ϊ��3CO32-+3Br2=5Br-+BrO3-+3CO2����
�ڴӢٳ�������Һ����ĺ������ߣ����ֱ������Ʒ�ɱ��ߣ�������Ҫ��һ��Ũ���壬������Ũ�ȣ�
�ʴ�Ϊ�������壬���Br2��Ũ�ȣ�
���¶ȹ���ˮ�������������к���ˮ�֣��¶ȹ����岻����ȫ���������ʵͣ�
�ʴ�Ϊ���¶ȹ������Խ�Br2�������������¶ȹ����ֻὫ������ˮ���������
�ʴ�Ϊ���ᾧ��
�������ӽ���Ĥֻ��������ͨ���������Ӻ����岻��ͨ������ʯī������ⱥ���Ȼ���ʱ�������������ӷŵ�������������������ͨ�������ӽ���Ĥ�����������������������������������ϲ�����ը����������������Һ��Ӧ�����Ȼ��ơ��������ƶ�������ȡ���������Ʋ�����
�ʴ�Ϊ����ֹH2��Cl2������Ӧ����������ը����ֹCl2�����ɵ�NaOH��Һ��Ӧ��ʹ�ռ��Ʒ�����ȣ�
��2���ٺ�ˮ�к��к�ˮ�к��϶�Mg2+��Ca2+�������ӣ����ʱ���������������������ӣ��������������Ӻ�þ���ӷ�Ӧ����Mg��OH��2��Ca��OH��2�ȳ����Ӷ����������ӽ���Ĥ���Ӷ���������ͨ�������µ�������ʧ�ܣ�
�ʴ�Ϊ����ˮ�к��϶�Mg2+��Ca2+�������ӣ����ʱ�����Mg��OH��2��Ca��OH��2�ȳ����Ӷ����������ӽ���Ĥ��
���ڵ�������������ˮʾ��ͼ������������������������������������������Ũ�ȱ�С����������Ũ�Ȳ�������Ũ�ȴ��ΪŨˮ������Ũˮ��B���ų�����ˮ��A���ų���
�ʴ�Ϊ����ˮ��
��3���ٴ�����̼���ƣ����巴Ӧ��BrO3-���ɣ���Ӧ�����ӷ���ʽΪ��3CO32-+3Br2=5Br-+BrO3-+3CO2����
�ʴ�Ϊ��3CO32-+3Br2=5Br-+BrO3-+3CO2����
�ڴӢٳ�������Һ����ĺ������ߣ����ֱ������Ʒ�ɱ��ߣ�������Ҫ��һ��Ũ���壬������Ũ�ȣ�
�ʴ�Ϊ�������壬���Br2��Ũ�ȣ�
���¶ȹ���ˮ�������������к���ˮ�֣��¶ȹ����岻����ȫ���������ʵͣ�
�ʴ�Ϊ���¶ȹ������Խ�Br2�������������¶ȹ����ֻὫ������ˮ���������
���������⿼�麣ˮ���ۺ����á���ˮɹ�Ρ���������������ˮ���Ӻ�ˮ�������ԭ������Ŀ�Ѷ��еȣ���ȷͼƬ�ں�����������Ϊ�Ȿ��ؼ�����2�����������ڵ缫�Ϸŵ磬�����ų���ˮΪ��ˮ���Ӷ�ʵ�ֺ�ˮ������

��ϰ��ϵ�д�
�����Ŀ
�������ӷ���ʽ��д��ȷ���ǣ�������
| A��ͭƬ������������Һ��Cu+Ag+=Cu2++Ag |
| B���������ᷴӦ��2Fe+6H+=2Fe3++3H2�� |
| C������������Һ�м�������������Һ�����ԣ�2H++SO42-+Ba2++2OH-=BaSO4��+2H2O |
| D������ʯ��ˮ�������ϣ�Ca��OH��2+2H+=Ca2++2H2O |
�����£�ȡpH������2������ʹ����100mL�ֱ�ϡ��10�����ٷֱ����0.03gп�ۣ�����ͬ�����³�ַ�Ӧ���й�������ȷ���ǣ�������
| A��������п��Ӧ�ų��������� |
| B��������п��Ӧ�ų��������� |
| C��������п��Ӧ�����ʴ� |
| D������ʹ���ֱ���п��Ӧ����һ���� |
������Ԫ��W��X��Y��Z��ԭ��������������������Ӳ����ƻ�ˮ�ĵ���ƽ����ǣ�������
| A��W- |
| B��X+ |
| C��Y3+ |
| D��Z2- |
��ʯ����һ���°����ʣ��ǡ�¹�ص���Լ���������Ƶ�46�ֻ�ѧƷ֮һ���仯ѧʽΪNa2Fe5Si8O22��OH��2����ʯ����ϡ������Һ����ʱ����ԭ����ֻ��NO������˵������ȷ���ǣ�������
| A����ʯ����һ�ֹ����β��� |
| B����ʯ���к���һ������ʯӢ���� |
| C����ʯ�Ļ�ѧ��ɿɱ�ʾΪNa2O?3FeO?Fe2O3?8SiO2?H2O |
| D��1 mol��ʯ���ܽ���1 mol HNO3��ϡ���ỹԭ |