��Ŀ����
���з�Ӧ������ˮ�ⷴӦ��ˮ�ⷽ��ʽ����ȷ���ǣ�������
��HCl+H2O?H3O++Cl-
��AlCl3+3H2O?Al��OH��3��+3HCl
��Na2CO3+2H2O?H2CO3+2NaOH
��̼��������Һ��HCO3-+H2O?CO32-+H3O+
��NH4Cl����D2O��NH4++D2O?NH3?D2O+H+
��HS-��ˮ��HS-+H2O?S2-+H2O+
�߽�����FeCl3��Һ������ڵ�ˮ�У�FeCl3+3 H2O?Fe��OH��3�����壩+3HCl��
��HCl+H2O?H3O++Cl-
��AlCl3+3H2O?Al��OH��3��+3HCl
��Na2CO3+2H2O?H2CO3+2NaOH
��̼��������Һ��HCO3-+H2O?CO32-+H3O+
��NH4Cl����D2O��NH4++D2O?NH3?D2O+H+
��HS-��ˮ��HS-+H2O?S2-+H2O+
�߽�����FeCl3��Һ������ڵ�ˮ�У�FeCl3+3 H2O?Fe��OH��3�����壩+3HCl��
| A���٢ڢۢ� | B���٢ڢ� |
| C���ڢۢܢݢ� | D��ȫ�� |
���㣺����ˮ���ԭ��,��ѧ����ʽ����д
ר�⣺
��������HClΪǿ����ʣ���ȫ���룻
��������ˮ��Ϊ���淴Ӧ���������ɳ�����
��̼�������ˮ��ֲ����У��Ե�һ��Ϊ����
��̼���������ˮ������̼�
��笠�����ˮ������һˮ�ϰ���
��HS-��ˮ�����������
�߱���FeCl3��Һ������ڵ�ˮ��ˮ�����ɽ��壮
��������ˮ��Ϊ���淴Ӧ���������ɳ�����
��̼�������ˮ��ֲ����У��Ե�һ��Ϊ����
��̼���������ˮ������̼�
��笠�����ˮ������һˮ�ϰ���
��HS-��ˮ�����������
�߱���FeCl3��Һ������ڵ�ˮ��ˮ�����ɽ��壮
���
�⣺��HCl�ĵ��뷽��ʽΪHCl+H2O�TH3O++Cl-���ʴ���
��ˮ�ⷴӦΪAlCl3+3H2O?Al��OH��3+3HCl���ʴ���
��ˮ�ⷴӦΪNa2CO3+H2O?NaHCO3+NaOH���ʴ���
��̼��������Һ��ˮ�����ӷ�ӦΪHCO3-+H2O?H2CO3+OH-���ʴ���
��NH4Cl����D2O�У�ˮ�����ӷ�ӦΪNH4++D2O?NH3?HDO+D+���ʴ���
��HS-��ˮ�����ӷ�ӦΪHS-+H2O?H2S++OH-���ʴ���
�߽�����FeCl3��Һ������ڵ�ˮ�У�ˮ�ⷴӦΪFeCl3+3 H2O
Fe��OH��3�����壩+3HCl���ʴ���
��ѡD��
��ˮ�ⷴӦΪAlCl3+3H2O?Al��OH��3+3HCl���ʴ���
��ˮ�ⷴӦΪNa2CO3+H2O?NaHCO3+NaOH���ʴ���
��̼��������Һ��ˮ�����ӷ�ӦΪHCO3-+H2O?H2CO3+OH-���ʴ���
��NH4Cl����D2O�У�ˮ�����ӷ�ӦΪNH4++D2O?NH3?HDO+D+���ʴ���
��HS-��ˮ�����ӷ�ӦΪHS-+H2O?H2S++OH-���ʴ���
�߽�����FeCl3��Һ������ڵ�ˮ�У�ˮ�ⷴӦΪFeCl3+3 H2O
| ||
��ѡD��
���������⿼�黯ѧ���Ϊ��Ƶ���㣬���յ��롢����ˮ���Ϊ���Ĺؼ���ע�������ˮ������𡢶�Ԫ���������ˮ��ֲ����м�ˮ��̶ȣ���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
����������ȷ���ǣ�������
| A��48g O3���庬��6.02��1023��O3���� |
| B��5.6g Fe��������ϡ���ᷴӦת�Ƶ���0.3 mol |
| C��0.5mol?L-1CuCl2��Һ�к���3.01��1023��Cu2+ |
| D����״���£�33.6LH2O����9.03��1023��H2O���� |
��NA��ʾ�����ӵ�������ֵ�����������в���ȷ���ǣ�������
| A����������ΪNA��NO2��CO2��������к��е���ԭ����Ϊ2NA |
| B��0.5molO3��11.2LO2�����ķ�����һ����� |
| C��28g��ϩ��C2H4���ͻ����飨C4H8���Ļ�������к��е�̼ԭ����Ϊ2NA |
| D��500mL 1mol/LNa2SO4��Һ�к�Na+��ĿΪNA |
 ��ͼ��ʾ�������̶�������������������ƶ���M��N���������о�������Ӧ��A��g��+3B��g��?2C��g����H=-192kJ?mol-1����M��N�У��ֱ�ͨ��x mol A��y mol B�Ļ�����壬��ʼM��N�ݻ���ͬ�������¶Ȳ��䣮����˵����ȷ���ǣ�������
��ͼ��ʾ�������̶�������������������ƶ���M��N���������о�������Ӧ��A��g��+3B��g��?2C��g����H=-192kJ?mol-1����M��N�У��ֱ�ͨ��x mol A��y mol B�Ļ�����壬��ʼM��N�ݻ���ͬ�������¶Ȳ��䣮����˵����ȷ���ǣ�������| A����ƽ��ʱA�������������е����������ȣ���xһ������y |
| B����x��y=1��2����ƽ��ʱ��M�е�ת���ʣ�A��B |
| C����x��y=1��3����M�зų�����172.8 kJʱ��A��ת����Ϊ90% |
| D����x=1.4��y=1��N�дﵽƽ��ʱ���Ϊ2L��CΪ0.4mol����Ӧ��ʼʱN�����Ϊ2.6L |
 ��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺
��B��ͬ���ڵ�һ��������С��Ԫ�أ�C��������������ɵ����ӣ�E����Χ�����Ų�ʽΪ3d64s2���ش��������⣺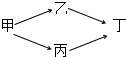 �ס��ҡ��������������ĺ��������������ж�����һ����һ�����ϵĹ����ţ�ͨ������ת����ϵ���ɼ���ȡ������������Է���������Ϊ���������ҵ���Է��������ܴ��ڱ����ش��������⣺
�ס��ҡ��������������ĺ��������������ж�����һ����һ�����ϵĹ����ţ�ͨ������ת����ϵ���ɼ���ȡ������������Է���������Ϊ���������ҵ���Է��������ܴ��ڱ����ش��������⣺