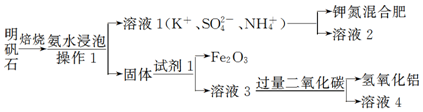
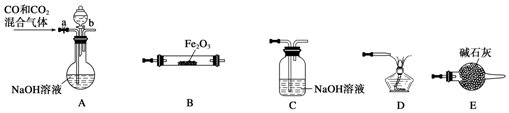
��Ŀ����
8������һ�ֳ����Ļ�ԭ������ͬ�����·ֽ���ﲻͬ��60-300��ʱ����Cu�Ƚ��������·ֽ�Ļ�����ͼ��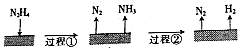
��֪��200��ʱ��
��3N2H4��g���TN2��g��+4NH3��g����H1=-32.9kJ•mol-1��
��N2H4��g��+H2��g���T2NH3��g����H2=-41.8kJ•mol-1��
����˵������ȷ���ǣ�������
| A�� | �����ڹ��ۻ����� | |
| B�� | ͼʾ���̢١��ڶ������ȷ�Ӧ | |
| C�� | ��Ӧ�����������뻹ԭ�������ʵ���֮��Ϊ2��1 | |
| D�� | 200��ʱ���·ֽ���Ȼ�ѧ����ʽΪ��N2H4��g���TN2��g��+2H2��g����H�T+50.7kJ•mol-1 |
���� A��ֻ���й��ۼ��Ļ��������ڹ��ۻ����
B�����̢���N2H4�ֽ�����N2��NH3�������Ȼ�ѧ����ʽ�С�H�жϣ�
C�����ϼ۽��͵õ���Ϊ�����������ϼ�����ʧ����Ϊ��ԭ����
D�����ݸ�˹���ɣ���I��-2����II����N2H4��g���TN2��g��+2H2��g���ݴ˼��㣮
��� �⣺A��ֻ���й��ۼ��Ļ��������ڹ��ۻ����N2H4�к��й��ۼ�����N2H4���ڹ��ۻ������A��ȷ��
B�����̢���N2H4�ֽ�����N2��NH3����֪�Ȼ�ѧ����ʽI�С�HΪ��ֵ������ͼʾ���̢�Ϊ���ȷ�Ӧ����B����
C��3N2H4��g���TN2��g��+4NH3��g����Ӧ�У�4��Nԭ�ӵĻ��ϼ۽��͵õ��ӣ�2��Nԭ�ӵĻ��ϼ�����ʧ���ӣ������������뻹ԭ�������ʵ���֮��Ϊ2��1����C��ȷ��
D�����ݸ�˹���ɣ���I��-2����II����N2H4��g���TN2��g��+2H2��g����H�T-32.9kJ•mol-1-2����-41.8kJ•mol-1��=+50.7kJ•mol-1����D��ȷ��
��ѡB��
���� ���⿼�����Ȼ�ѧ����ʽ�ļ��㡢��˹���ɵļ���Ӧ�á�������ԭ��Ӧ����Ŀ�Ѷ��еȣ�ע����ո�˹���ɵ�Ӧ�÷����Լ�������ԭ��Ӧ���йظ�������ڿ���ѧ���ķ��������ͼ���������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�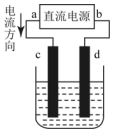
| A�� | c��������ԭ��Ӧ | B�� | ÿ����6.4gCu��ת�Ƶ���0.2mol | ||
| C�� | ������Ӧʽ��Cu2++2e��Cu | D�� | ����Һ�е�����d����c |
| ѡ�� | �ı������ | �����淴Ӧ���ʱ仯 | ��ƽ���ԭƽ��Ƚ� |
| A | ����ѹǿ | �����淴Ӧ���ʶ������� | Z��Ũ�ȼ�С |
| B | ��������Z | �淴Ӧ�������� | X������������� |
| C | ��������Z | v���棩��v������ | Z��Ũ�ȼ�С |
| D | �����¶� | �淴Ӧ�������������� ����Ӧ���������� | Z������������ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | PH3�ǷǼ��Է��� | |
| B�� | PH3��������δ�ɼ��ĵ��Ӷ� | |
| C�� | PH3�е�P-H���ļ��Ա�NH3��N-H���ļ����� | |
| D�� | PH3�����е�P-H���ǷǼ��Լ� |
| A�� | ��ⱥ���Ȼ�þ��Һ��2Cl-+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$H2��+Cl2��+2OH- | |
| B�� | Fe��OH��3��������Fe��OH��3+3H+�TFe3++3H2O | |
| C�� | NaAlO2��Һ��AlO2-��ˮ�⣺AlO2-+2H2O�TAl��OH��3+OH- | |
| D�� | ��KAl��SO4��2��Һ�мӹ�����Ba��OH��2��Һ��Al3++2SO42-+2Ba2++4OH-�TAlO2-+2BaSO4��+2H2O |
| A�� | ��a=bʱ�������������ܷ�ӦΪ��Ba2++2OH-+CO2�TBaCO3��+H2O | |
| B�� | ��3a��2bʱ���������ӷ�ӦΪ��OH-+CO2�THCO3- | |
| C�� | ��2a=bʱ�������������ܷ�ӦΪ��Ba2++3OH-+2CO2�TBaCO3��+H2O+HCO3- | |
| D�� | 2a��b��3aʱ����Һ��n��HCO3-��=��2b-3a��mol |
| A�� | 75% | B�� | 50% | C�� | 5% | D�� | 12.5% |
| A�� | ������ܶȲ���ʱ��˵���ﵽ��ƽ��״̬ | |
| B�� | ����������X����H��� | |
| C�� | ����һ�����Ķ������壬ƽ�������ƶ� | |
| D�� | ����������Y����Ӧ���ʼӿ� |