��Ŀ����
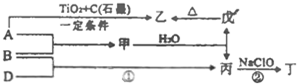 ��֪��A��B��DΪ��ѧ�����ĵ��ʣ��ס��ҡ�����������Ϊ������Ԫ����ɵĻ�������У�����һ����ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���壻����һ�ָ���ȼ�ϣ������Ԫ�������ͬ��1mol�������в�ͬԭ�ӵ���Ŀ��Ϊ1��2���Һ���18mol���ӣ�����һ��������ˮ�İ�ɫ��״���ʣ�������ǿ�ᷴӦ��Ҳ����ǿ�Ӧ�����о�ˮ���ã������ʼ��ת����ϵ��ͼ��ʾ��ijЩ��������ȥ����
��֪��A��B��DΪ��ѧ�����ĵ��ʣ��ס��ҡ�����������Ϊ������Ԫ����ɵĻ�������У�����һ����ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���壻����һ�ָ���ȼ�ϣ������Ԫ�������ͬ��1mol�������в�ͬԭ�ӵ���Ŀ��Ϊ1��2���Һ���18mol���ӣ�����һ��������ˮ�İ�ɫ��״���ʣ�������ǿ�ᷴӦ��Ҳ����ǿ�Ӧ�����о�ˮ���ã������ʼ��ת����ϵ��ͼ��ʾ��ijЩ��������ȥ���� ��ش�
��1������B�����Ԫ�������ڱ��е�λ����
��2������ҵĻ�ѧ����ʽΪ
��3�������������Ļ�ѧ��������
a�����Ӽ�������������������b�����Թ��ۼ���������������������c���Ǽ��Թ��ۼ�
��4����Ӧ�ٵĻ�ѧ����ʽΪ
��5����Ӧ���У�0.5mol��NaClO�μӷ�Ӧʱ��ת��1mol�����ӣ��仯ѧ����ʽΪ��
��6��һ�������£�A��TiO2��C��ʯī����Ӧֻ�����Һ�̼���ѣ�TiC�������߾�ΪijЩ���½ṹ�մɵ���Ҫ�ɷ֣���֪���÷�Ӧ����1mol��ʱ�ų�536kJ���������Ȼ�ѧ����ʽΪ
���㣺������ƶ�
ר�⣺�ƶ���
������A��B��DΪ��ѧ�����ĵ��ʣ��ס��ҡ�����������Ϊ������Ԫ����ɵĻ��������һ����ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���壬���ΪNH3������һ�ָ���ȼ�ϣ������Ԫ�������ͬ��1mol�������в�ͬԭ�ӵ���Ŀ��Ϊ1��2���Һ���18mol���ӣ���ΪN2H4������һ��������ˮ�İ�ɫ��״���ʣ�������ǿ�ᷴӦҲ����ǿ�Ӧ�����о�ˮ���ã�����ΪAl��OH��3��
���ת����ϵͼ��֪����Ϊ��������AΪAl��BΪN2����Ϊ��������DΪH2��Ȼ�������ʵ����ʼ���ѧ���������
���ת����ϵͼ��֪����Ϊ��������AΪAl��BΪN2����Ϊ��������DΪH2��Ȼ�������ʵ����ʼ���ѧ���������
���
�⣺A��B��DΪ��ѧ�����ĵ��ʣ��ס��ҡ�����������Ϊ������Ԫ����ɵĻ��������һ����ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���壬���ΪNH3������һ�ָ���ȼ�ϣ������Ԫ�������ͬ��1mol�������в�ͬԭ�ӵ���Ŀ��Ϊ1��2���Һ���18mol���ӣ���ΪN2H4������һ��������ˮ�İ�ɫ��״���ʣ�������ǿ�ᷴӦҲ����ǿ�Ӧ�����о�ˮ���ã�����ΪAl��OH��3��
���ת����ϵͼ��֪����Ϊ��������AΪAl��BΪN2����Ϊ��������DΪH2��
��1�����B��Ԫ��ΪNԪ�أ�λ��Ԫ�����ڱ��е�2���ڵڢ�A�壬�ʴ�Ϊ����2���ڣ���VA�壻
��2����ΪAl��OH��3�����ȷֽ�����������������ʽΪ2Al��OH��3
Al2O3+3H2O���ʴ�Ϊ��2Al��OH��3
Al2O3+3H2O��
��3�����ǰ����������е�ԭ�Ӻ���ԭ��֮����ڼ��Թ��ۼ����ʴ�Ϊ��b��
��4����Ӧ��Ϊ�ϳɰ��ķ�Ӧ������ʽΪN2+3H2
2NH3���ʴ�Ϊ��N2+3H2
2NH3��
��5����Ӧ��Ϊ������NaClO�ķ�Ӧ��0.5molNaClO�μӷ�Ӧʱ��ת��1mol���ӣ���ClԪ����+1�۽���Ϊ-1�ۣ��÷�Ӧ����NaCl��ͬʱ����N2H4��ˮ����÷�ӦΪ2NH3+NaClO�TN2H4+NaCl+H2O��
�ʴ�Ϊ��2NH3+NaClO�TN2H4+NaCl+H2O��
��6����-�������£�A��TiO2��C��ʯī����Ӧֻ�����Һ�̼���ѣ�TiC������Al��TiO2��C��Ӧ����Al2O3��TiC������1molAl2O3ʱ�ų�536kJ������������2molAl2O3ʱ�ų�536kJ��2=1072�����������Ȼ�ѧ��Ӧ����ʽΪ4Al��s��+3TiO2��s��+3C��s��ʯī���T2Al2O3��s��+3TiC��s����H=-1072 kJ/mol��
�ʴ�Ϊ��4Al��s��+3TiO2��s��+3C��s��ʯī���T2Al2O3��s��+3TiC��s����H=-1072 kJ/mol��
���ת����ϵͼ��֪����Ϊ��������AΪAl��BΪN2����Ϊ��������DΪH2��
��1�����B��Ԫ��ΪNԪ�أ�λ��Ԫ�����ڱ��е�2���ڵڢ�A�壬�ʴ�Ϊ����2���ڣ���VA�壻
��2����ΪAl��OH��3�����ȷֽ�����������������ʽΪ2Al��OH��3
| ||
| ||
��3�����ǰ����������е�ԭ�Ӻ���ԭ��֮����ڼ��Թ��ۼ����ʴ�Ϊ��b��
��4����Ӧ��Ϊ�ϳɰ��ķ�Ӧ������ʽΪN2+3H2
| ���¸�ѹ |
| ���� |
| ���¸�ѹ |
| ���� |
��5����Ӧ��Ϊ������NaClO�ķ�Ӧ��0.5molNaClO�μӷ�Ӧʱ��ת��1mol���ӣ���ClԪ����+1�۽���Ϊ-1�ۣ��÷�Ӧ����NaCl��ͬʱ����N2H4��ˮ����÷�ӦΪ2NH3+NaClO�TN2H4+NaCl+H2O��
�ʴ�Ϊ��2NH3+NaClO�TN2H4+NaCl+H2O��
��6����-�������£�A��TiO2��C��ʯī����Ӧֻ�����Һ�̼���ѣ�TiC������Al��TiO2��C��Ӧ����Al2O3��TiC������1molAl2O3ʱ�ų�536kJ������������2molAl2O3ʱ�ų�536kJ��2=1072�����������Ȼ�ѧ��Ӧ����ʽΪ4Al��s��+3TiO2��s��+3C��s��ʯī���T2Al2O3��s��+3TiC��s����H=-1072 kJ/mol��
�ʴ�Ϊ��4Al��s��+3TiO2��s��+3C��s��ʯī���T2Al2O3��s��+3TiC��s����H=-1072 kJ/mol��
���������⿼��������ƶϣ�Ϊ��Ƶ���㣬������ѧ���ķ��������Ŀ��飬���ʵ��ƶ��ǽ����Ĺؼ���������Ϊ������ͻ�ƿڣ�����Ϥ��ѧ�����ʹ������ɣ���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
0.1mol NaHCO3���������
| A��0.2 mol Na+ |
| B��0.05 mol CO |
| C��6.02��1023�� O |
| D��0.1 mol H |

