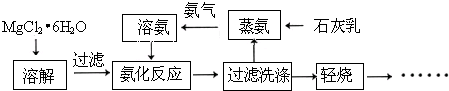
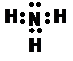��Ŀ����
��������FeCl3��Һ�μӵķ�Ӧ�У���Fe3+ˮ���йصķ�Ӧ��
��FeCl3��Һ��Cu�ķ�Ӧ �ڽ�FeCl3��Һ�������ɣ����������յõ�Fe2O3 ��FeCl3��Һ��KI�ķ�Ӧ �ܱ���FeCl3��Һ�����ˮ���Ʊ�Fe(OH)3���� ��FeCl3��Һ��H2S�ķ�Ӧ��FeCl3��Һ��NaHCO3��Һ�ķ�Ӧ ������FeCl3��Һ�����һ����������
| A���٢ܢ� | B���ڢۢݢ� | C���ڢܢޢ� | D���٢ڢۢܢݢޢ� |
C
������ȷ�𰸣�C
ˮ�ⷽ��Ϊ;Fe3����3H2O Fe(OH)3��3H��
Fe(OH)3��3H��
����Fe3+ˮ���أ�FeCl3�������� ����Fe3+ˮ���йأ���FeCl3��Һ���ȣ�HCl�ӷ���Fe(OH)3���պ�ֽ⣬���յõ�Fe2O3 ����Fe3+ˮ���أ�FeCl3�������� ����Fe3+ˮ���йأ�����FeCl3��Һ�����ˮ��ˮ�⣬�Ʊ�Fe(OH)3���� ����Fe3+ˮ���أ�FeCl3��Һ������������Fe3+ˮ���йأ�FeCl3ˮ������ԣ�NaHCO3ˮ��ʼ��ԣ���ٽ� ����Fe3+ˮ���йأ�����FeCl3��Һ�����һ���������ᣬ����ˮ�⣬��ֹ����������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��1����4�֣������й�ʵ�����������˵������ȷ����
| A����pH��ֽ������ˮʪ�����ij��Һ��pH |
| B����ͭƬ����Ƭ������һ�����ϡ�����У�ͭƬ����������� |
| C���ζ���ϴ��������ˮ��ϴ����ע���Һ�����к͵ζ�ʵ�� |
| D�����������Һ���������������ͨ��һ����HCl����ʱ���������Һ�ɻָ�����ԭ��һ�� |
F��ʵ����������FeCl3��Һʱ���Ƚ�FeCl3����һ������Ũ�����У��ټ�����ˮϡ��������Ũ��
��2����6�֣�ijͬѧ�ڰ�����ʦ����ʵ���ҵĻ�ѧ�Լ�ʱ������һʢ����ɫ��Һ���Լ�ƿ����ǩ����(����ͼ)��������������յ�֪ʶ���Ը��Լ�������ʲô���ʵ���Һ�������룬�����ʵ�������֤��

�ٲ��룺�����Լ�������_____________________��
�ڼ�����֤��ʵ�鷽���� ��