��Ŀ����
2��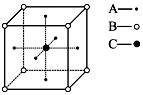 ��1�����Ȼ���������Ϊ���壬�۵�282�棬�е�315�棬��300��������������������ˮ��Ҳ���������ѡ���ͪ���л��ܼ����ݴ��ж����Ȼ�����������Ϊ���Ӿ��壮
��1�����Ȼ���������Ϊ���壬�۵�282�棬�е�315�棬��300��������������������ˮ��Ҳ���������ѡ���ͪ���л��ܼ����ݴ��ж����Ȼ�����������Ϊ���Ӿ��壮��2����CH2=CH2����CH��CH����
 ����CH3-CH3�����ַ�����̼ԭ�Ӳ�ȡsp2�ӻ����Ǣ٢ۣ�����ţ���
����CH3-CH3�����ַ�����̼ԭ�Ӳ�ȡsp2�ӻ����Ǣ٢ۣ�����ţ�����3����ͼΪij�����һ������ʾ��ͼ���þ�����A��B��C���ֻ���������ɣ���þ���Ļ�ѧʽΪCBA3��
���� ��1�����Ӿ����۷е�ϵͣ�
��2����CH2=CH2������̼ԭ�Ӳ�ȡsp2�ӻ�����CH��CH������̼ԭ�Ӳ�ȡsp�ӻ����� ������̼ԭ�Ӳ�ȡsp2�ӻ�����CH3-CH3������̼ԭ�Ӳ�ȡsp3�ӻ���
������̼ԭ�Ӳ�ȡsp2�ӻ�����CH3-CH3������̼ԭ�Ӳ�ȡsp3�ӻ���
��3�����ݾ���ͼ��֪��Aԭ�ӷֲ�������������������ϣ����Ծ����к���Aԭ�ӵ���ĿΪ$6��\frac{1}{2}$=3��Bԭ�ӷֲ���������İ˸������ϣ����Ծ�������Bԭ����ĿΪ$8��\frac{1}{8}$=1��Cԭ��λ������������ģ���ĿΪ1�������ھ�����ABC����ԭ�ӵĸ�����Ϊ3��1��1���ݴ˴��⣮
��� �⣺��1�����Ӿ����۷е�ϵͣ������ʵ��۷е�ϵͣ�����Ϊ���Ӿ��壬�ʴ�Ϊ�����Ӿ��壻
��2����CH2=CH2������̼ԭ�Ӳ�ȡsp2�ӻ�����CH��CH������̼ԭ�Ӳ�ȡsp�ӻ����� ������̼ԭ�Ӳ�ȡsp2�ӻ�����CH3-CH3������̼ԭ�Ӳ�ȡsp3�ӻ������Ԣ٢�̼ԭ���Dz�ȡsp2�ӻ���
������̼ԭ�Ӳ�ȡsp2�ӻ�����CH3-CH3������̼ԭ�Ӳ�ȡsp3�ӻ������Ԣ٢�̼ԭ���Dz�ȡsp2�ӻ���
�ʴ�Ϊ���٢ۣ�
��3�����ݾ���ͼ��֪��Aԭ�ӷֲ�������������������ϣ����Ծ����к���Aԭ�ӵ���ĿΪ$6��\frac{1}{2}$=3��Bԭ�ӷֲ���������İ˸������ϣ����Ծ�������Bԭ����ĿΪ$8��\frac{1}{8}$=1��Cԭ��λ������������ģ���ĿΪ1�������ھ�����ABC����ԭ�ӵĸ�����Ϊ3��1��1�����Ի�ѧʽΪ��CBA3���ʴ�Ϊ��CBA3��
���� ���⿼�����ʽṹ�����ʣ���Ϥ���ʵ��������ʼ����ʵijɼ����ɽ���ѶȽϴ�ע��ѧ������Ϣ�Ĵ������ϺõĿ���ѧ���������⡢��������������

| ѡ�� | ���ʣ�������Ϊ���ʣ� | �����Լ� | ���鷽�� |
| A | CO2��SO2�� | ����NaHSO3��Һ | ���ѳ����ʵ���������ͨ��Ʒ����Һ |
| B | NaHCO3��Һ��Na2CO3�� | ����CO2 | ȡ������Һ������Ca ��OH��2��Һ���۲����� |
| C | NO��NO2�� | ˮ | �۲���ɫ |
| D | ���飨��ϩ�� | ����KMnO4��Һ | ���ѳ����ʵ���������ͨ����ˮ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ǿ�������Һ�ĵ�������һ�������������Һ��ǿ | |
| B�� | ��������ǿ����ʣ�������������� | |
| C�� | ������������ʣ�ͭ��ǿ����� | |
| D�� | ��������ǿ����ʣ����ᱵ��������� |
��ȡ20.0mL����Һ������25.0mL 4.00mol•L-1NaOH��Һ���а�ɫ���������ݼ���ζ���壮���ˡ�ϴ�ӡ�����ó���1.16g���ٽ���Һϡ����100mL�������Һ��c��OH����Ϊ0.20mol•L-1��
����ȡ20.0mL����Һ������������AgNO3��Һ�����ɰ�ɫ����11.48g��
�ɴ˿ɵó�����ԭ��Һ��ɵ���ȷ�����ǣ�������
| A�� | һ������Mg2+��Al3+��Cl�������ܺ���Na+������NH4+ | |
| B�� | һ������Mg2+��Cl��������NH4+�����ܺ���Na+��Al3+ | |
| C�� | c ��Cl���� Ϊ 4.00 mol•L-1��c��Al3+��Ϊ5.00 mol•L-1 | |
| D�� | c ��Mg2+�� Ϊ 1.00 mol•L-1��c��Na+��Ϊ 0.5mol•L-1 |

| A�� | ̼����������ų�����ҺpH��� | |
| B�� | a��������b�Ǹ��� | |
| C�� | �������е������������Ӵ�a������b�� | |
| D�� | a���Ϸ�����������Ӧ |
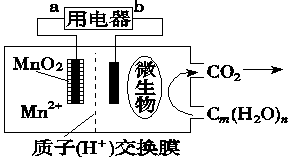
| A�� | ��ع���ʱ��������b����a | |
| B�� | ������ӦʽΪ��MnO2+4H++2e-�TMn2++2H2O | |
| C�� | �ŵ�����У�H+�Ӹ��������������� | |
| D�� | �������ڵ缫���ŵ�ʱ������ԭ��Ӧ |
��������
| A�� | AlO2- | B�� | Fe3+ | C�� | HCO3- | D�� | NH4+ |
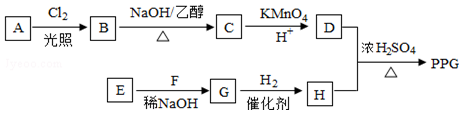
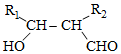
 �����Ը��������Һ�з�Ӧ����HOOCCH2CH2CH2CH2COOH
�����Ը��������Һ�з�Ӧ����HOOCCH2CH2CH2CH2COOH ��
�� ��
��
 ��
��