��Ŀ����
15��ʵ��������Ҫ����180mL1mol•L-1SrCl2��Һ������ʵ����Ʒ��ʵ���������ȷ���ǣ�������| ѡ�� | ����ƿ�ݻ� | �������� | ʵ����� |
| A | 180mL | SrCl2��28.6g | ����180mLˮ |
| B | 200mL | SrCl2��31.8g | ���200mL��Һ |
| C | 250mL | SrCl2��39.8g | ���250mL��Һ |
| D | 250mL | SrCl2•2H2O��39.8g | ���250mL��Һ |
| A�� | A | B�� | B | C�� | C | D�� | D |
���� ����180mL1mol•L-1SrCl2��Һ��ʵ����û��180mL������ƿ����Ӧѡ��250mL����ƿ��������Һ�����Ϊ250mL����ҪSrCl2���ʵ�������SrCl2��������ʵ���������m=cVM��������SrCl2��������SrCl2������������ݴ��жϣ�ע��250mL����Һ������������ܼ������Ϊ250mL��
��� �⣺A������180mL1mol•L-1SrCl2��Һ��ʵ����û��180mL������ƿ����Ӧѡ��250mL����ƿ����A����
B������180mL1mol•L-1SrCl2��Һ��Ӧѡ��250mL����ƿ����B����
C������180mL1mol•L-1SrCl2��Һ��ʵ����û��180mL������ƿ����Ӧѡ��250mL����ƿ��������Һ�����Ϊ250mL����ҪSrCl2�����ʵ�����Ϊ��1mol/L��159g/mol��0.25L=39.8g����C��ȷ��
D������180mL1mol•L-1SrCl2��Һ��ʵ����û��180mL������ƿ����Ӧѡ��250mL����ƿ��������Һ�����Ϊ250mL����ҪSrCl2�����ʵ�����Ϊ��1mol/L��195g/mol��0.25L=48.8g����D����
��ѡ��C��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ���ǽ���ؼ���ע������ƿ���ѡ����Ŀ�ѶȲ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�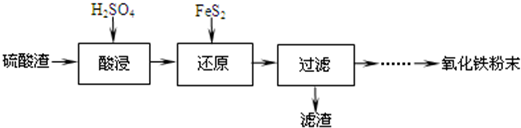
��1���������������Ҫ�ʵ�������Ŀ���ǣ�
��������Ľ����ʣ�������Fe3+��ˮ�⣮
��2������ԭ���ǽ�Fe3+ת��ΪFe2+��ͬʱFeS2������ΪSO42-���÷�Ӧ�����ӷ���ʽΪ14Fe3++FeS2+8H2O=15Fe2++2SO42-+16H+��
��3��Ϊ�ⶨ��������������Һ��Fe3+�����Կ��Ƽ���FeS2������ʵ�鲽��Ϊ��
ȷ��ȡһ���������������Һ����ƿ�У�����HCl���Թ���SnCl2���ټ�HgCl2��ȥ������SnCl2���Զ�����������Ϊָʾ������K2Cr2O7����Һ�ζ����йط�Ӧ����ʽ���£�
2Fe3++Sn2++6Cl-�T2Fe2++SnCl62-��
Sn2++4Cl-+2HgCl2�TSnCl62-+Hg2Cl2����
6Fe2++Cr2O72-+14H+�T6Fe3++2Cr3++7H2O��
����SnCl2����������ⶨ��Fe3+��ƫ�ͣ��ƫ�ߡ�����ƫ�͡��������䡱����ͬ����
��������HgCl2����ⶨ��Fe3+��ƫ�ߣ�
��4����֪����������������������ʽ����ʱ��Һ��pH������
| ������ | Fe��OH��3 | Al��OH��3 | Fe��OH��2 | Mg��OH��2 | Mn��OH��2 |
| ��ʼ���� | 2.7 | 3.8 | 7.5 | 9.4 | 8.3 |
| ��ȫ���� | 3.2 | 5.2 | 9.7 | 12.4 | 9.8 |
a������������Һ�м�����������Ը��������Һ�����裻
b���������μ�NaOH��Һ��������ҺΪpHΪ3.2��3.8��
c�����룬ϴ�ӣ�
d����ɣ���ĥ��
������ƽ��ʹ�����룩����ʱ�����������������λ�÷ŵߵ���
������Ͳ��ȡҺ��ʱ�����Ӷ���
����Һת�Ƶ�����ƿ���ձ���������δ������ˮϴ��
��ת����Һǰ����ƿ������������ˮ
�ݶ���ʱ����������ƿ�Ŀ̶���
���ݺ�ҡ�ȣ�����Һ�潵�ͣ��ֲ�������ˮ�����´ﵽ�̶��ߣ�
| A�� | �٢ۢݢ� | B�� | �٢ڢݢ� | C�� | �ڢۢܢ� | D�� | �ۢܢݢ� |
 ˮú������Ҫȼ�Ϻͻ���ԭ�ϣ�����ˮ����ͨ�����ȵ�̿���Ƶã�
ˮú������Ҫȼ�Ϻͻ���ԭ�ϣ�����ˮ����ͨ�����ȵ�̿���Ƶã�C ��s��+H2O��g��?CO ��g��+H2 ��g����H=+131.3kJ•mol-1
��1���÷�Ӧ��ƽ�ⳣ��K���¶ȵ����߶���������С�����䣩��
��2��������Ӧ�ﵽƽ�������H2O��g����������C ��s�� �����ʵ�����С������С�����䣩��
��3������˵��һ�����ж����Ϸ�Ӧ��ƽ�����BC
A������1mol H2O��g��ͬʱ����1mol H2
B�������ڻ�����������������
C�����º���ʱ�����������ѹǿ����
D��H2O��g����CO ��g����H2 ��g�����������Ũ�ȱ�Ϊ1��1��1
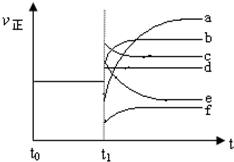
��4��������Ӧ��t0ʱ�̴ﵽƽ�⣬��t1ʱ�̸ı�ijһ����������Ӧ���ʣ���������ʱ��ı仯����ͼ��ʾ�������Ӧ�ı�ţ�
�����������b��
�ڽ����¶�f��
��5��һ���¶��£����������о�������������Ӧ����������̿�������������ʵ����ʵ���Ũ�ȼ����淴Ӧ���ʹ�ϵ���±���ʾ������д���Т٢���Ӧ�Ŀո�
| ������� | c��H2O��/mol•L-1 | c��CO��/mol•L-1 | c��H2��/mol•L-1 | �����������Ƚ� |
| I | 0.06 | 0.60 | 0.10 | ����=���� |
| �� | 0.12 | 0.20 | 0.60�� | ����=���� |
| �� | 0.10 | 0.20 | 0.40 | ����_�ڣ����� |
| A�� | B2H6��g��+3O2��g���TB2O3��s��+3H2O��g����H=-2165kJ•mol-1 | |
| B�� | B2H6��g��+3O2��g���TB2O3��s��+3H2O��g����H=-2033kJ•mol-1 | |
| C�� | B2H6��g��+3O2��g���TB2O3+3H2O��H=-2165kJ•mol-1 | |
| D�� | B2H6��g��+O2��g���TB2O3��s��+H2O��l����H=-2165kJ•mol-1 |
| A�� | HCl+Na2CO3��HCl+NaHCO3 | B�� | BaCl2+Na2SO4��Ba��OH��2+NaHSO4 | ||
| C�� | KOH+HCl��Ba��OH��2+H2SO4 | D�� | ʯ��ʯ�����ᡢʯ��ʯ������ |
| A�� | NaOH��Һ�е���CuSO4��Һ��OH-+Cu2+=Cu��OH��2�� | |
| B�� | ��ȩ����������ӦCH3CHO+2[Ag��NH3��2]++2OH-$\stackrel{��}{��}$CH3COO-+NH4+2Ag��+3NH3+H2O | |
| C�� | ������̼������Һ��Ӧ��CO32-+2H+=H2O+CO2�� | |
| D�� | �������е���AgNO3��Һ��Ag++Br-=AgBr�� |
