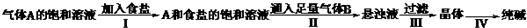МвДҝДЪИЭ
ОТ№ъ»ҜС§јТәоөВ°сёщҫЭNaHCO3ИЬҪв¶ИұИNaClЎўNa2CO3ЎўNH4HCO3ЎўNH4Cl¶јРЎөДРФЦКЈ¬ФЛУГCO2 +NH3 +H2O+NaClЈҪNaHCO3Ўэ+NH4ClөД·ҙУҰФӯАнЦЖұёҙҝјоЎЈПВГжКЗФЪКөСйКТҪшРРДЈДвКөСйөДЙъІъБчіМКҫТвНјЈә
 |
ФтПВБРРрКцҙнОуөДКЗ
AЈ®AЖшМеКЗNH3Ј¬BЖшМеКЗCO2
BЈ®°Сҙҝјој°өЪўуІҪөГөҪөДҫ§МеУлДіР©№ММеЛбРФОпЦКЈЁИзҫЖКҜЛбЈ©»мәПҝЙЦЖөГ·ўҪН·Ы
CЈ®ҙҝјоҝЙ№г·әөШУГУЪІЈБ§ЎўЦЖФнЎўФмЦҪЎў·ДЦҜөИ№ӨТөЦР
DЈ®өЪўфІҪІЩЧчКЗҪ«ҫ§МеИЬУЪЛ®әујУИИЎўХф·ўЎўҪбҫ§
D

Б·П°ІбПөБРҙр°ё
 ФД¶БҝміөПөБРҙр°ё
ФД¶БҝміөПөБРҙр°ё
Па№ШМвДҝ
ОТ№ъ»ҜС§јТәоөВ°сёщҫЭNaHCO3ИЬҪв¶ИұИNaClЎўNa2CO3ЎўNH4HCO3ЎўNH4Cl¶јРЎөДРФЦКЈ¬ФЛУГCO2+NH3+H2O+NaCl=NaHCO3Ўэ+NH4ClөД·ҙУҰФӯАнЦЖұёҙҝјоЈ®ПВГжКЗФЪКөСйКТҪшРРДЈДвКөСйөДЙъІъБчіМКҫТвНјЈә
ЖшМеAөДұҘәНИЬТә
AәНКіСОөДұҘәНИЬТә
РьЧЗТә
ҫ§Ме
ҙҝјо
ФтПВБРРрКцҙнОуөДКЗЈЁЎЎЎЎЈ©
ЖшМеAөДұҘәНИЬТә
| јУИлКіСО |
| ўс |
| НЁИлЧгБҝЖшМеB |
| ўт |
| №эВЛ |
| ўу |
| ўф |
ФтПВБРРрКцҙнОуөДКЗЈЁЎЎЎЎЈ©
| AЎўAЖшМеКЗCO2Ј¬BЖшМеКЗNH3 |
| BЎўөЪўуІҪөГөҪөДҫ§МеКЗ·ўҪН·ЫөДЦчТӘіЙ·Ц |
| CЎўөЪўуІҪІЩЧчУГөҪөДЦчТӘІЈБ§ТЗЖчКЗЙХұӯЎўВ©¶·ЎўІЈБ§°ф |
| DЎўөЪўфІҪІЩЧчөДЦчТӘ№эіМУРИЬҪвЎўХф·ўЎўҪбҫ§ |