��Ŀ����
ijѧ������18.4mol/L ��Ũ��������0.4mol/L ���� 250mL���Իش��������⣺
��1��ͨ����������Ũ����V mL��V ��ֵΪ ��
��2����������Ʒ��ѡ��ʵ���������������Ʒ ������ţ���ͬ����
���ձ� �ڲ����� ��10mL ��Ͳ ��100mL ��Ͳ ��500mL ����ƿ ��250mL ����ƿ �߹��ƿ ��������ƽ
��ѡ�����������⣬��ȱ�ٵ������� ��
��3�����в����У�������� ��������ţ���ͬ��
������Ͳ��ȡV mL Ũ���� ������Ͳ�м�����������ˮ�����ò����������������ϡ�ͺ����Һת������ƿ�� ��Ȼ��С�ĵ�������ƿ�м�ˮ���̶�1-2cm�� ��ƽ�Ӱ�Һ�涨�ݢް�����ƿƿ������������ҡ��
��4�����д�������У���ʹ�����Ƶ���Һ��H2SO4 �����ʵ���Ũ��ƫ�͵��� ��
�ٽ�ϡ�ͺ������ת��������ƿ�к�δϴ���ձ��Ͳ�����
�ڽ��ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ��
�۶��ݲ�������ʹ��Һ��Һ���������ƿ�̶��ߣ���ʱ�����������Һ��������ʹ��Һ��Һ��������ƿ�̶�������
�ܶ���ʱ��������ƿ�̶���
�ݶ���ʱ��������ƿ�̶��ߣ�
��1��ͨ����������Ũ����V mL��V ��ֵΪ
��2����������Ʒ��ѡ��ʵ���������������Ʒ
���ձ� �ڲ����� ��10mL ��Ͳ ��100mL ��Ͳ ��500mL ����ƿ ��250mL ����ƿ �߹��ƿ ��������ƽ
��ѡ�����������⣬��ȱ�ٵ�������
��3����������������
������Ͳ��ȡV mL Ũ���� ������Ͳ�м�����������ˮ�����ò����������������ϡ�ͺ����Һת������ƿ�� ��Ȼ��С�ĵ�������ƿ�м�ˮ���̶�1-2cm�� ��ƽ�Ӱ�Һ�涨�ݢް�����ƿƿ������������ҡ��
��4�����д�������У���ʹ�����Ƶ���Һ��H2SO4 �����ʵ���Ũ��ƫ�͵���
�ٽ�ϡ�ͺ������ת��������ƿ�к�δϴ���ձ��Ͳ�����
�ڽ��ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ��
�۶��ݲ�������ʹ��Һ��Һ���������ƿ�̶��ߣ���ʱ�����������Һ��������ʹ��Һ��Һ��������ƿ�̶�������
�ܶ���ʱ��������ƿ�̶���
�ݶ���ʱ��������ƿ�̶��ߣ�
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺ʵ����
��������1��������Һϡ���������ʵ����ʵ�������������ҪŨ����������
��2����������250mL 0.4mol/L��������Һ�IJ���ѡ��������Ȼ���жϻ�ȱ�ٵ��������ƣ�
��3����������һ�����ʵ���Ũ�ȵ���Һ����ȷ�������������жϣ�
��4������c=
�ɵã�һ�����ʵ���Ũ����Һ���Ƶ����������ʵ����ʵ���n����Һ�����V����ģ�������ʱ���ؼ�Ҫ�����ƹ���������n��V�����ı仯����n������ֵС����V������ֵ��ʱ������ʹ������ҺŨ��ƫС����n������ֵ��V������ֵСʱ������ʹ������ҺŨ��ƫ��
��2����������250mL 0.4mol/L��������Һ�IJ���ѡ��������Ȼ���жϻ�ȱ�ٵ��������ƣ�
��3����������һ�����ʵ���Ũ�ȵ���Һ����ȷ�������������жϣ�
��4������c=
| n |
| V |
���
�⣺��1������250mL 0.4mol/L��������Һ�����У�������������ʵ������䣬������ҪŨ��������Ϊ��
��0.054L=5.4mL������V=5.4��
�ʴ�Ϊ��5.4��
��2������250mL 0.4mol/L��ϡ����IJ���Ϊ���������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װƿ����ǩ����Ҫѡ�õ�����Ϊ��10mL��Ͳ���ձ�����������250ml����ƿ����ͷ�ιܣ�ѡ������Ҫѡ�õ�����Ϊ�����ձ����ڲ���������10mL��Ͳ����250mL ����ƿ����ȱ�ٽ�ͷ�ιܣ�
�ʴ�Ϊ���٢ڢۢޣ���ͷ�ιܣ�
��3��������Ͳ��ȡV mL Ũ���������10mL��Ͳ��ȡ5.4mLŨ���ᣬ�ò����������ʢ���ȷ��
������Ͳ�м�����������ˮ�����ò��������裻ϡ��Ũ����Ӧ�����ձ��н��У�����ʹ����Ͳϡ�ͣ��ʢڴ���
��������ϡ�ͺ����Һת������ƿ�У�Ũ������ϡ�����зų���������Ҫ��ȴ�����º���ת�Ƶ�����ƿ�У��ʢ۴���
�ܶ���ʱ����С�ĵ�������ƿ�м�ˮ���̶�1-2cm�����ô��ں������ʢ���ȷ��
������ý�ͷ�ιܶ���ʱ��Ҫƽ�Ӱ�Һ�涨�ݣ��ʢ���ȷ��
�ް�����ƿƿ������������ҡ�ȣ���������ƿ��Һ���������ô��ں������ʢ���ȷ��
�ʴ�Ϊ���ڢۣ�
��4���ٽ�ϡ�ͺ������ת��������ƿ�к�δϴ���ձ��Ͳ��������������Ƶ���Һ�����ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ��ʢ���ȷ��
�ڽ��ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�⣬�������Ƶ���Һ�����ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ��ʢ���ȷ��
�۶��ݲ�������ʹ��Һ��Һ���������ƿ�̶��ߣ��������Ƶ���Һ���ƫ����ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ܶ���ʱ��������ƿ�̶��ߣ����¼��������ˮ���ƫ�����Ƶ���Һ���ƫ��Ũ�ȵ�ƫ�ͣ��ʢ���ȷ��
�ݶ���ʱ��������ƿ�̶��ߣ����¼��������ˮ���ƫС�����Ƶ���Һ���ƫС����Һ��Ũ��ƫ�ߣ��ʢݴ���
�ʴ�Ϊ���٢ڢۢܣ�
| 0.4mol/L��0.25L |
| 18.4mol/L |
�ʴ�Ϊ��5.4��
��2������250mL 0.4mol/L��ϡ����IJ���Ϊ���������ȡ��ϡ�͡���ȴ����Һ�����ݡ�ҡ�ȡ�װƿ����ǩ����Ҫѡ�õ�����Ϊ��10mL��Ͳ���ձ�����������250ml����ƿ����ͷ�ιܣ�ѡ������Ҫѡ�õ�����Ϊ�����ձ����ڲ���������10mL��Ͳ����250mL ����ƿ����ȱ�ٽ�ͷ�ιܣ�
�ʴ�Ϊ���٢ڢۢޣ���ͷ�ιܣ�
��3��������Ͳ��ȡV mL Ũ���������10mL��Ͳ��ȡ5.4mLŨ���ᣬ�ò����������ʢ���ȷ��
������Ͳ�м�����������ˮ�����ò��������裻ϡ��Ũ����Ӧ�����ձ��н��У�����ʹ����Ͳϡ�ͣ��ʢڴ���
��������ϡ�ͺ����Һת������ƿ�У�Ũ������ϡ�����зų���������Ҫ��ȴ�����º���ת�Ƶ�����ƿ�У��ʢ۴���
�ܶ���ʱ����С�ĵ�������ƿ�м�ˮ���̶�1-2cm�����ô��ں������ʢ���ȷ��
������ý�ͷ�ιܶ���ʱ��Ҫƽ�Ӱ�Һ�涨�ݣ��ʢ���ȷ��
�ް�����ƿƿ������������ҡ�ȣ���������ƿ��Һ���������ô��ں������ʢ���ȷ��
�ʴ�Ϊ���ڢۣ�
��4���ٽ�ϡ�ͺ������ת��������ƿ�к�δϴ���ձ��Ͳ��������������Ƶ���Һ�����ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ��ʢ���ȷ��
�ڽ��ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�⣬�������Ƶ���Һ�����ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ��ʢ���ȷ��
�۶��ݲ�������ʹ��Һ��Һ���������ƿ�̶��ߣ��������Ƶ���Һ���ƫ����ҺŨ��ƫ�ͣ��ʢ���ȷ��
�ܶ���ʱ��������ƿ�̶��ߣ����¼��������ˮ���ƫ�����Ƶ���Һ���ƫ��Ũ�ȵ�ƫ�ͣ��ʢ���ȷ��
�ݶ���ʱ��������ƿ�̶��ߣ����¼��������ˮ���ƫС�����Ƶ���Һ���ƫС����Һ��Ũ��ƫ�ߣ��ʢݴ���
�ʴ�Ϊ���٢ڢۢܣ�
���������⿼��������һ�����ʵ���Ũ�ȵ���Һ�ķ��������������ǿ��ע������ԣ����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ����Ҫ��ѧ����������һ�����ʵ���Ũ�ȵ���Һ�ķ�����������ѵ���������������Ҫ��ȷ�������ķ����뼼�ɣ���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �����Ļ�������ҵϵ�д�
�����Ļ�������ҵϵ�д�
�����Ŀ
���й�������Ҫ�����������ǣ�������
��N2��2N ��2Cl��Cl2
������ʯ��ʯ����ʯ�� ��Zn+2HCl�TZnCl2+H2��
��2KOH+H2SO4�T2H2O+K2SO4 ��2NH4Cl+Ba��OH��2�TBaCl2+2NH3��+2H2O
��ʳ�︯�� ��C+H2O��g��
CO+H2��
��N2��2N ��2Cl��Cl2
������ʯ��ʯ����ʯ�� ��Zn+2HCl�TZnCl2+H2��
��2KOH+H2SO4�T2H2O+K2SO4 ��2NH4Cl+Ba��OH��2�TBaCl2+2NH3��+2H2O
��ʳ�︯�� ��C+H2O��g��
| ||
| A���٢ۢ� | B���٢ۢޢ� |
| C���ۢܢޢ� | D���ڢۢܢݢ� |
���и�����������Һ��ķ�Ӧ��������ͬһ���ӷ���ʽ��ʾ���ǣ�������
| A��HCl��Na2CO3 HCl��NaHCO3 |
| B��AgNO3��HCl AgNO3��H2SO4 |
| C��BaCl2��Na2SO4 Ba��OH��2��CuSO4 |
| D��KOH��CuCl2 Ba��OH��2��CuCl2 |
 ��ͼ�����ڼ��л���������Ʊ������롢�ᴿ�����ļ���װ�ã�����ݸ�װ�ûش��������⣺
��ͼ�����ڼ��л���������Ʊ������롢�ᴿ�����ļ���װ�ã�����ݸ�װ�ûش��������⣺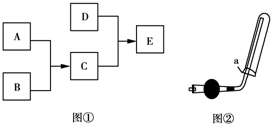 ��֪A��B��C��DΪ���壬����AΪ����ɫ��D��������ˮ���γɵ���Һ��ʹ��̪��죬����֮���ת����ϵ��ͼ����ʾ��
��֪A��B��C��DΪ���壬����AΪ����ɫ��D��������ˮ���γɵ���Һ��ʹ��̪��죬����֮���ת����ϵ��ͼ����ʾ��