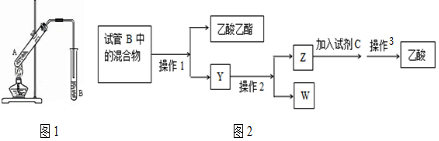
��Ŀ����
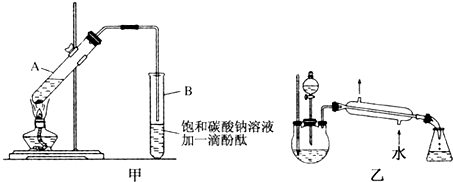
6��ʵ������������������Ҫװ����ͼA��ʾ����Ҫ�������a�Թ��а�2��3��2�����������Ũ���ᡢ�Ҵ�������Ļ����ڰ�Aͼ����װ�ã�ʹ����������������ͨ��b�Թ���ʢ�ı���̼������Һ�����뼸�η�̪��Һ���У���С�����a�Թ��еĻ��Һ���ܵ�b�Թ����ռ���Լ2mL����ʱֹͣ���ȣ�����b�Թܲ�������Ȼ���ô�����Һ��ֲ㣻�ݷ��������������������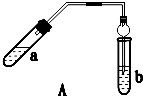
��ش��������⣺
��1��������пɹ۲쵽b�Թ�����ϸС������ð����д���÷�Ӧ�����ӷ���ʽ��2CH3COOH+CO32-=2CH3COO-+H2O+CO2����
��2��Aװ����ʹ�����ιܳ������������⣬��һ��Ҫ�����Ƿ�ֹ������������з���������������ʹ�õ�һ�������Ƿ�Һ©����
��3��Ϊ֤��Ũ�����ڸ÷�Ӧ�����˴�������ˮ�������ã�ijͬѧ������ͼA��ʾװ�ý���������4��ʵ�飮ʵ�鿪ʼ���þƾ�����3min���ټ���ʹ֮����3min��ʵ�����������С�Թ�b�ٲ��л���ĺ�ȣ�ʵ���¼���£�
| ʵ�� ��� | �Թ�a���Լ� | �Թ�b���Լ� | ����л���ĺ��/cm |
| A | 3 mL�Ҵ���2 mL���ᡢ1mL 18mol•L-1 Ũ���� | ����Na2CO3��Һ | 5.0 |
| B | 3 mL�Ҵ���2 mL���� | 0.1 | |
| C | 3 mL�Ҵ���2 mL���ᡢ6 mL 3mol•L-1 H2SO4 | 1.2 | |
| D | 3 mL�Ҵ���2 mL���ᡢ���� | 1.2 |
�ڷ���ʵ��AC����ʵ���ţ������ݣ������Ʋ��ŨH2SO4����ˮ����������������IJ��ʣ�Ũ�������ˮ���ܹ���������������ʵ�ԭ����Ũ�����������������Ӧ�����ɵ�ˮ��������������Ũ��ʹƽ�����������������ķ����ƶ���
�ۼ���������������������IJ��ʣ���ʵ�鷢���¶ȹ������������IJ��ʷ������ͣ����ܵ�ԭ���Ǵ������ᡢ�Ҵ�δ����Ӧ�����뷴Ӧ��ϵ�����¶ȹ��߷���������Ӧ����
�ܷ�����������������ϴ�ӣ�Ϊ�˸�������������ѡ�õĸ����ΪB������ĸ����
A��P2O5 B����ˮNa2SO4 C����ʯ�� D��NaOH���壮
���� ��1��������̼���Ʒ�Ӧ���ɶ�����̼�������ƺ�ˮ��
��2�����ᡢ�Ҵ�������̼������Һ���ᵼ��װ���������С������������������������ˮ��Һ�ֲ㣬���뻥�����ܵ�Һ���ȡ��Һ������
��3����ͨ������ʵ�飬֤��H+��������Ӧ���д����ã�Ӧ��֤������Ũ����������ʵ�����ȣ�
�ڶԱ�Ũ���ᡢϡ���������������������Ķ��٣�˵��Ũ����Ũ�������ˮ���ܹ���������������ʣ�����ƽ���ƶ�ԭ��������
�����ᡢ�Ҵ��ӷ���Ҳ���ܷ�����������Ӧ��
���������������ԡ����������¾����Է���ˮ�⣬Ӧѡ�����Ը������
��� �⣺��1��������̼���Ʒ�Ӧ���ɶ�����̼�������ƺ�ˮ����ѧ����ʽ��2CH3COOH+CO32-=2CH3COO-+H2O+CO2����
�ʴ�Ϊ��2CH3COOH+CO32-=2CH3COO-+H2O+CO2����
��2�����ᡢ�Ҵ�������̼������Һ���ᵼ��װ���������С�����������������θ���ܻ����Է�ֹ����������������ˮ��Һ�ֲ㣬���뻥�����ܵ�Һ���ȡ��Һ��������������Ϊ��Һ©����
�ʴ�Ϊ����ֹ��������Һ©����
��3����ͨ������ʵ�飬֤��H+��������Ӧ���д����ã�Ӧ��֤������Ũ����������ʵ�����ȣ���HCl��Ũ��Ϊ3mol/L��2=6mol/L���������Ϊ6mL��
�ʴ�Ϊ��6��6��
�ڴ���ΪŨ���������л����ȱ�3mol•L-1 H2SO4ʱ�����л����Ⱥ�˵��Ũ����Ũ�������ˮ���ܹ���������������ʣ�ԭ���ǣ�Ũ�����������������Ӧ�����ɵ�ˮ��������������Ũ��ʹƽ�����������������ķ����ƶ���
�ʴ�Ϊ��AC��Ũ�����������������Ӧ�����ɵ�ˮ��������������Ũ��ʹƽ�����������������ķ����ƶ���
�����ᡢ�Ҵ��ӷ����¶ȹ��ߴ������ᡢ�Ҵ�δ����Ӧ�����뷴Ӧ��ϵ�����¶ȹ��߷���������Ӧ�����²��ʽ��ͣ�
�ʴ�Ϊ���������ᡢ�Ҵ�δ����Ӧ�����뷴Ӧ��ϵ���¶ȹ��߷���������Ӧ��
���������������ԡ����������¾����Է���ˮ�⣬Ӧѡ�����Ը������
��ѡ��B��
���� ���⿼���л����Ʊ�ʵ�顢̽��ʵ�飬��3����ע�����ÿ��Ʊ������������ۺϿ���ѧ��ʵ������������������֪ʶǨ�������������Ѷ��еȣ�

 ����ѵ����ֱͨ�п�����ϵ�д�
����ѵ����ֱͨ�п�����ϵ�д� һ���㶨ϵ�д�
һ���㶨ϵ�д� ��У��ҵ��ϵ�д�
��У��ҵ��ϵ�д� ���ɶ���ܲ��¿�ֱͨ��Уϵ�д�
���ɶ���ܲ��¿�ֱͨ��Уϵ�д�| ���� | �۵㣨�棩 | �е㣨�棩 | �ܶȣ�g•cm-3�� |
| �Ҵ� | -117.3 | 78.5 | 0.79 |
| ���� | 16.6 | 117.9 | 1.05 |
| �������� | -83.6 | 77.5 | 0.90 |
| Ũ���� | - | 338.0 | 1.84 |
����30mL�Ĵ��Թ�A�а������2��3��3����Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ�ͼ�����Ӻ�װ�ã�װ�����������ã�����С����ȼ���װ�л��Һ�Ĵ��Թ�5��10min��
�۴��Թ�B�ռ���һ���������ֹͣ���ȣ������Թ�B��������Ȼ���ô��ֲ㣮
�ܷ�������������㣬ϴ�ӡ����
�������ĿҪ��ش��������⣺
��1�����Ƹû��Һ����Ҫ��������Ϊ��һ��30mL�Ĵ��Թ���ע��3mL�Ҵ����ٷֱ�����2mLŨ���ᡢ3mL���ᣨ�Ҵ���Ũ����ļ���˳�ɻ��������ӱ����Թ�ʹ֮��Ͼ��ȣ�
��2�����������ҪС����ȼ��ȣ�����Ҫԭ���Ƿ�Ӧ���Ҵ�������ķе�ϵͣ����ô����ȣ���Ӧ�������������������ʧ�������¶ȹ��߿��ܷ�������ĸ���Ӧ��
��3��ָ����������۲쵽�������Թ�B�е�Һ��ֳ��������㣬�ϲ���ɫ���²�Ϊ��ɫҺ�壬���²�Һ��ĺ�ɫ��dz��
����������������һ���ñ���ʳ��ˮ���Ȼ�����Һϴ�ӣ���ͨ��ϴ����Ҫ��ȥ̼���ƺ��Ҵ��������ƣ����ʣ�Ϊ�˸�������������ѡ�õĸ����ΪB����ѡ����ĸ����
A��P2O5 B����ˮNa2SO4
C����ʯ�� D��NaOH����
��4��ij��ѧ����С���������ͼ����ʾ����ȡ����������װ�ã�ͼ�еIJ���װ����ȥ������ͼ��װ����ȣ�ͼ��װ�õ���Ҫ�ŵ��Т��������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����ķ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������
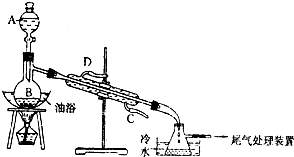 ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A��ʢ��Ũ���ᣬB��ʢ����ˮ�Ҵ��ͱ����ᣮ
ij����С����Ƶ�ʵ������ȡ����������װ����ͼ��ʾ��A��ʢ��Ũ���ᣬB��ʢ����ˮ�Ҵ��ͱ����ᣮ��֪����ˮ�Ȼ��ƿ����Ҵ��γ�������ˮ��CaCl2•6C2H5OH��
���й��л���ķе������ʾ��
| �Լ� | ���� | �Ҵ� | ���� | �������� |
| �е㣨�棩 | 34.7 | 78.5 | 118 | 77.1 |
 CH3CO18OCH2CH3+H2O��
CH3CO18OCH2CH3+H2O����2���ù����Ҵ�����ҪĿ��������һ�ַ�Ӧ�������������Ӧ������У�
��3�������ܵ���Ҫ�����ǽ�������������ˮӦ�������ܵ�C���C����D�����˽��룮
��4����ƿ���ռ�����Һ�����Ҫ�ɷ�������������Ϊ�˵õ��Ƚϴ����ĸ����ʣ����ñ���Na2CO3��Һ�Դֲ�Ʒ����ϴ�ӣ���Ŀ���dz�ȥ���������е�������Ҵ��������NaOHŨ��Һ����Na2CO3��Һ������ĺ���ǵ�����������ˮ�⣮
��5����ƿ��Һ�徭����̼������Һϴ�Ӻ�����ˮ�Ȼ��ƣ���ȥ�Ҵ����ټ��루�˿մ�����ѡ����ѡ��C��Ȼ����������ռ�77�����ҵ���ּ��ɣ�
A������������B����ʯ��C����ˮ������D����ʯ�ң�
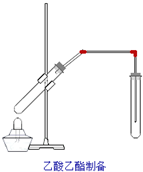 ��ͼ�������Թ����ȼ���2mL 95%���Ҵ�������ҡ���»�������3mLŨ���ᣬ�ټ���2mL���ᣬ���ҡ�ȣ������Թ��м���5mL����Na2CO3��Һ����ͼ���Ӻ�װ�ã��þƾ��ƶ����Թ�С�����3��5min���ô����ȣ����۲쵽���Թ�������������ʱֹͣʵ��
��ͼ�������Թ����ȼ���2mL 95%���Ҵ�������ҡ���»�������3mLŨ���ᣬ�ټ���2mL���ᣬ���ҡ�ȣ������Թ��м���5mL����Na2CO3��Һ����ͼ���Ӻ�װ�ã��þƾ��ƶ����Թ�С�����3��5min���ô����ȣ����۲쵽���Թ�������������ʱֹͣʵ��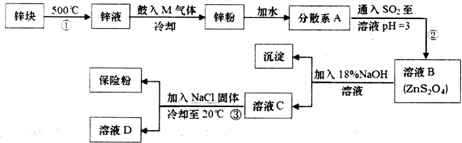
 ����ɢϵAΪ����Һ�����Һ�������塱������Һ������
����ɢϵAΪ����Һ�����Һ�������塱������Һ������