��Ŀ����
�л���A����ѧʽΪC4H8O3����һ�������µ������У���Ũ��������£�����ˮ������ʹ��ˮ��ɫ��ֻ��һ�ֽṹ��ʽ���л���B����Ũ��������£��ֱܷ����Ҵ������ᷴӦ�����л���C��D����Ũ��������£��������ɷ���ʽΪC8H12O4����Ԫ��״�л���E���������й�����һ����ȷ�ģ�������
| A���л���A������Ϊ��-�ǻ����� |
| B���л���B�Ľṹ��ʽ����ΪCH3CH=CHCOOH |
| C���л���C��D��ͬ���칹�� |
| D����״�л���E�Ľṹֻ��һ�� |
���㣺�л���Ľṹ������
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������Ũ��������£��ֱܷ����Ҵ������ᷴӦ�����л���C��D����֪���л����к���-OH��-COOH��
��Ũ��������£�����ˮ������ʹ��ˮ��ɫ��ֻ��һ�ֽṹ��ʽ���л���B����֪-OH��λ�ϵ�Cԭ������Hԭ�ӣ���ֻ����һ��ϩ����
��Ũ��������£��������ɷ���ʽΪC8H12O4����Ԫ��״�л���E����A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH���Դ˽����⣮
��Ũ��������£�����ˮ������ʹ��ˮ��ɫ��ֻ��һ�ֽṹ��ʽ���л���B����֪-OH��λ�ϵ�Cԭ������Hԭ�ӣ���ֻ����һ��ϩ����
��Ũ��������£��������ɷ���ʽΪC8H12O4����Ԫ��״�л���E����A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH���Դ˽����⣮
���
�⣺��Ũ��������£��ֱܷ����Ҵ������ᷴӦ�����л���C��D����֪���л����к���-OH��-COOH��
��Ũ��������£�����ˮ������ʹ��ˮ��ɫ��ֻ��һ�ֽṹ��ʽ���л���B����֪-OH��λ�ϵ�Cԭ������Hԭ�ӣ���ֻ����һ��ϩ����
��Ũ��������£��������ɷ���ʽΪC8H12O4����Ԫ��״�л���E����A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH��
A��A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH������Ϊ��-�ǻ������2-��-��-�ǻ����ᣬ��A����
B����AΪCH3CH2CHOHCOOH����B����ΪCH3CH=CHCOOH����B��ȷ��
C��CH3CH2CHOHCOOH�����ǻ����Ȼ�����Ũ��������£��ֱܷ����Ҵ������ᷴӦ�����л���C��D����ʽ��ͬ������ͬ���칹�壬��C����
D��A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH������Ԫ��״�л���E����Ϊ ��
��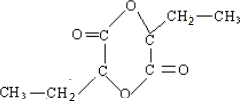 ����D����
����D����
��ѡB��
��Ũ��������£�����ˮ������ʹ��ˮ��ɫ��ֻ��һ�ֽṹ��ʽ���л���B����֪-OH��λ�ϵ�Cԭ������Hԭ�ӣ���ֻ����һ��ϩ����
��Ũ��������£��������ɷ���ʽΪC8H12O4����Ԫ��״�л���E����A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH��
A��A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH������Ϊ��-�ǻ������2-��-��-�ǻ����ᣬ��A����
B����AΪCH3CH2CHOHCOOH����B����ΪCH3CH=CHCOOH����B��ȷ��
C��CH3CH2CHOHCOOH�����ǻ����Ȼ�����Ũ��������£��ֱܷ����Ҵ������ᷴӦ�����л���C��D����ʽ��ͬ������ͬ���칹�壬��C����
D��A����ΪCH3CH2CHOHCOOH��CH3C��CH3��OHCOOH������Ԫ��״�л���E����Ϊ
 ��
�� ����D����
����D������ѡB��
���������⿼���л���Ľṹ�����ʣ�Ϊ��Ƶ���㣬������ѧ���ķ��������Ŀ��飬ע����������Ϣ�Լ������ŵ����ʣ���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��NAΪ����ӵ�������������������ȷ���ǣ�������
| A��1L 0.1mol/L��������Һ����������Ϊ0.2NA |
| B����״����22.4 L SO3 �к���ԭ����Ϊ3NA |
| C��1mol���������к���3NA�����ۼ� |
| D�������£�1molFe������Ũ���ᷴӦʧȥ3NA������ |
��NA���������ӵ�������ֵ������˵������ȷ���ǣ�������
| A����NA��ͭԭ�ӵ�CuO��Cu2S�Ļ��������Ϊ80.0 g |
| B����״���£�22.4 L������������ʱת�Ƶ�����Ϊ4NA |
| C��500 mL��2 mol?L-1̼������Һ�д���������һ������3NA |
| D��50 mL 18 mol?L-1Ũ����������п����Ӧ�����������������С��0.9NA |
�ڲ�ͬ�����·ֱ��N2��g��+3H2��g��?2NH3��g���ķ�Ӧ���ʣ����б�ʾ�÷�Ӧ���������ǣ�������
| A��v ��N2��=3 mol?L-1?min-1 |
| B��v ��NH3��=2 mol?L-1?s-1 |
| C��v ��H2��=2.5mol?L-1?s-1 |
| D��v ��NH3��=10mol?L-1?min-1 |
 ����һ�������¿ɱ������ɶ���ȩ������ij�л�������A��C6H8������һ��������������������Ӧ�����ɱ����л�������B��C6H12��������ij�������������л�������Aʱ�����ɱ���ȩ��OHC-CH2-CHO������
����һ�������¿ɱ������ɶ���ȩ������ij�л�������A��C6H8������һ��������������������Ӧ�����ɱ����л�������B��C6H12��������ij�������������л�������Aʱ�����ɱ���ȩ��OHC-CH2-CHO������