��Ŀ����
��Ի�ѧ��Ӧ�е������仯����������⡣
��1���ⶨϡ�����ϡ���������к��ȣ��к���Ϊ57.3 kJ.mor1����ʵ��װ�D��ͼ��ʾ��
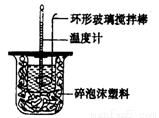
ij��ȤС���ʵ����ֵ���С��57.3 kJ/mol��ԭ�������________������ĸ����
A��ʵ��װ�D���¡�����Ч����
B����ȡ���Һ������¶ȼ�Ϊ�յ��¶�
C���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
D�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�δϴ�ӣ�ֱ�ӲⶨH2SO4 ��Һ���¶�
��2����������������ȡ��ҵԭ�����ᡣ��֪��
A��CH3COOH��l��+2O2��g��=2CO2��g��+2H2O��l�� ��H=��870.3kJ��mo1
B��C��s��+O2��g��=CO2��g�� ��H=��393.5kJ��mo1
C��H2��g��+1/2O2��g��=H2O��l�� ��H=��285.8kJ��mo1
����ͬ������CH3COOH��C��H2��ȫȼ��ʱ���ų�����������___________��
������������Ϣ����������Ӧ��2C��s��+2H2��g��+O2��g��=CH3COOH��l�� ��H= kJ/mol ��
��3����Cl2����ijЩ�����л���ʱ�����������HC1�����÷�ӦA����ʵ���ȵ�ѭ�����á�
��Ӧ A: 4HCl��O2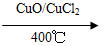 2Cl2��2H2O
2Cl2��2H2O
��1����֪����.��ӦA�У�4 molHCl���������ų�115.6 kJ��������

��.
��д���������·�ӦA���Ȼ�ѧ����ʽ��________________��
�Ͽ�1 mol H��O����Ͽ�1 mol H��Cl�������������ԼΪ________kJ��
̼��������ҹ���Ҫ�ĵ���Ʒ��֮һ���������������������ӷ���ʧ��Ϊ�˼�����������ȷ�����ʩ����������ⶨ�京������
��ijѧ�������һ���Բⶨ������̼������Ӳⶨ�������ķ���������Ʒ����Բ����ƿ�У�
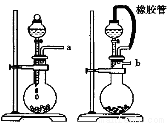
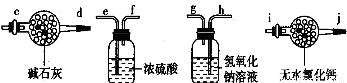
��1����ѡ���Ҫ��װ�ã���������������˳��Ϊ ��
��2����Һ©���е�Һ�����ʺϵ��� ��
A��ϡ���� B��ϡ���� C��Ũ���� D����������
��3����������װ�������� ��
����������гɷ��ǣ�NH4��2SO4��������ü�ȩ���ⶨ����������ȩ���ǻ��ڼ�ȩ��һ������������ã������൱�����ᣬ��ӦΪ2��NH4��2SO4+6HCHO=��CH2��6N4 +2H2SO4 + 6H2O,���ɵ��������������Ʊ���Һ�ζ����Ӷ��ⶨ���ĺ������������£�
��4���ò�������ȡ���壨NH4��2SO4��Ʒ0.6g���ձ��У�����Լ30mL����ˮ�ܽ⣬�������100mL��Һ���� �����ʽ����ʽ�����ζ���ȷȡ��20��00mL����Һ����ƿ�У�����18%���Լ�ȩ��Һ5mL������5min����1~2�� ָʾ������֪�ζ��յ��pHԼΪ8��8������Ũ��Ϊ0��08mol/L�������Ʊ���Һ�ζ����������±���
�ζ����� | �ζ�ǰ������mL�� | �ζ��������mL�� |
1 | 1��20 | 16��21 |
2 | 3��00 | 18��90 |
3 | 4��50 | 19��49 |
��ζ��յ�ʱ������Ϊ ���ɴ˿ɼ��������Ʒ�еĵ�����������Ϊ ��
��5���ڵζ�ʵ��������ֵζ��õļ�ʽ�ζ��ܲ��������ڳ��������ݣ��ζ���ʼʱ�����ݣ����ʵ��ⶨ�ĺ�������ʵ��ֵ ���ƫ��ƫС������Ӱ�족����
������ⶨ̼������еĺ�����ʱ��ʹ�ü�ȩ���Ƿ���� ����ǡ����������� ��


 22.4 Lһ����̼����ԭ����
22.4 Lһ����̼����ԭ���� Ũ�Ⱥ�10%NaOH��Һ��NaOH�����ʵ���Ũ��
Ũ�Ⱥ�10%NaOH��Һ��NaOH�����ʵ���Ũ�� 2CrO42-+2H+��������Һ��ɫ�ж��������
2CrO42-+2H+��������Һ��ɫ�ж�������� 2SO3��g�� ��H=-197kJ/mol�����ﵽ��ѧƽ��ʱ������˵������ȷ����
2SO3��g�� ��H=-197kJ/mol�����ﵽ��ѧƽ��ʱ������˵������ȷ����