��Ŀ����
ij��ɫ��Һ��ֻ��������NH4+��K+��Al3+��Mg2+��HCO3-��Cl-��I-��MnO4-��SO42-�������еļ������ӣ���������ɣ��ֽ�������ʵ�飺
��ȡl0mL����Һ���Թ��еμ�������Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�
��ȡ������Ӧ�����Һ������AgNO3��Һδ������������
����ȡl0mL����Һ���Թ��У��μ�NaOH��Һ������ɫ�����ң�������NaOH�����ʵ���Ϊ0.03molʱ�����������ﵽ������μ�NaOH��Һ�����ȣ���ʼ������������ռ���������������ɱ����Ϊ0.224L�������ȫ���ݳ�������������ȫ�ܽ⣮�����ƶ���ȷ���ǣ�������
��ȡl0mL����Һ���Թ��еμ�������Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�
��ȡ������Ӧ�����Һ������AgNO3��Һδ������������
����ȡl0mL����Һ���Թ��У��μ�NaOH��Һ������ɫ�����ң�������NaOH�����ʵ���Ϊ0.03molʱ�����������ﵽ������μ�NaOH��Һ�����ȣ���ʼ������������ռ���������������ɱ����Ϊ0.224L�������ȫ���ݳ�������������ȫ�ܽ⣮�����ƶ���ȷ���ǣ�������
| A���϶���K+��Al3+��Mg2+��SO42- |
| B���϶���K+��NH4+��Al3+��SO42- |
| C���϶�û��K+��HCO3-��MnO4- |
| D���϶�û��K+��NH4+��Cl- |
���㣺���������ӵļ���,���������ӵļ���
ר�⣺���ʼ��������
��������Һ��ɫ��˵������MnO4-��
��ȡ10mL����Һ���Թ��еμ�Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�˵����ΪBaSO4��ԭ��Һ�к���SO42-��
������Һ�м���AgNO3��Һδ������������˵��������Cl-��I-��
����Ϊ���������������ֻ��NH4+��������þ����������������Һ��Al3+��������������Һ��Ӧ���ɳ���������ȫ�ܽ⣬��������̼����������ܷ���˫ˮ�ⷴӦ�����ܴ������棬�ʲ���HCO3-������������������0.01mol�ͱ���°���Ϊ0.224L������Ԫ���غ��֪��NH4+0.01mol��Al3+Ϊ0.01mol���ٸ��ݵ���غ����֪����K+����һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-��
��ȡ10mL����Һ���Թ��еμ�Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�˵����ΪBaSO4��ԭ��Һ�к���SO42-��
������Һ�м���AgNO3��Һδ������������˵��������Cl-��I-��
����Ϊ���������������ֻ��NH4+��������þ����������������Һ��Al3+��������������Һ��Ӧ���ɳ���������ȫ�ܽ⣬��������̼����������ܷ���˫ˮ�ⷴӦ�����ܴ������棬�ʲ���HCO3-������������������0.01mol�ͱ���°���Ϊ0.224L������Ԫ���غ��֪��NH4+0.01mol��Al3+Ϊ0.01mol���ٸ��ݵ���غ����֪����K+����һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-��
���
�⣺��Һ��ɫ��˵������MnO4-��
��ȡ10mL����Һ���Թ��еμ�Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�˵����ΪBaSO4��ԭ��Һ�к���SO42-��
������Һ�м���AgNO3��Һδ������������˵��������Cl-��I-��
����Ϊ���������������ֻ��NH4+��������þ����������������Һ��Al3+��������������Һ��Ӧ���ɳ���������ȫ�ܽ⣬��������̼����������ܷ���˫ˮ�ⷴӦ�����ܴ������棬�ʲ���HCO3-������������������0.01mol�ͱ���°���Ϊ0.224L������Ԫ���غ��֪��NH4+0.01mol��Al3+Ϊ0.01mol���ٸ��ݵ���غ����֪����K+��������������֪һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-��
A��������������֪��һ������þ���ӣ���A����
B��������������֪��һ�����е�����ΪK+��NH4+��Al3+��SO42-����B��ȷ��
C��һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-����C����
D���ɵ���غ��֪��һ����K+����D����
��ѡB��
��ȡ10mL����Һ���Թ��еμ�Ba��NO3��2��Һ����ϡ�����ữ����˵õ�0.03mol��ɫ�����ף�˵����ΪBaSO4��ԭ��Һ�к���SO42-��
������Һ�м���AgNO3��Һδ������������˵��������Cl-��I-��
����Ϊ���������������ֻ��NH4+��������þ����������������Һ��Al3+��������������Һ��Ӧ���ɳ���������ȫ�ܽ⣬��������̼����������ܷ���˫ˮ�ⷴӦ�����ܴ������棬�ʲ���HCO3-������������������0.01mol�ͱ���°���Ϊ0.224L������Ԫ���غ��֪��NH4+0.01mol��Al3+Ϊ0.01mol���ٸ��ݵ���غ����֪����K+��������������֪һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-��
A��������������֪��һ������þ���ӣ���A����
B��������������֪��һ�����е�����ΪK+��NH4+��Al3+��SO42-����B��ȷ��
C��һ�����е�����ΪK+��NH4+��Al3+��SO42-��һ�������е�����ΪMnO4-��Mg2+��Cl-��I-��HCO3-����C����
D���ɵ���غ��֪��һ����K+����D����
��ѡB��
���������⿼�����ʼ����ʵ�鷽������ƣ�Ϊ��Ƶ���㣬����ʵ������������۵Ĺ�ϵ�����������ӷ�ӦΪ���Ĺؼ���ע�����غ��Ӧ�ã���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
aLFe2��SO4��3��Һ�к�Fe3+bg������Һ��SO42-�����ʵ���Ũ��Ϊ��������mol/L��
A��
| ||
B��
| ||
C��
| ||
D��
|
����ͼʾ�����У�����ȷ���ǣ�������
A�� ���� |
B�� ����ʳ��ˮ |
C�� ��ȡʱ����Һ©�� |
D�� ������Һʱ��������ƿ�������µߵ���ҡ�� |
������ɫ��Ӧ����K+ʱ�������²��裬������ȷ�IJ���˳���ǣ�������
��պȡ����Һ �����ھƾ��ƻ��������� ������ɫ�ܲ����۲� ����ϡ����ϴ����˿��
��պȡ����Һ �����ھƾ��ƻ��������� ������ɫ�ܲ����۲� ����ϡ����ϴ����˿��
| A���ܢڢ٢ڢ� | B���٢ڢۢ� |
| C���ܢ٢ڢ� | D���ڢ٢ۢ� |
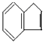 ��һ��̼�⻯�������һ��ͬ���칹��A��A�����е�̼ԭ�Ӳ�һ��ȫ��ͬһƽ���ϣ���A�����б�����ֻ��һ��ȡ������A�����±仯��ϵ��
��һ��̼�⻯�������һ��ͬ���칹��A��A�����е�̼ԭ�Ӳ�һ��ȫ��ͬһƽ���ϣ���A�����б�����ֻ��һ��ȡ������A�����±仯��ϵ��
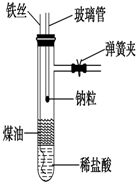 ��1����ͼ��ʾ��װ��������һ֧С�ľ�֧�Թ��м���2mL���ҵ�ϡ���ᣬ�ټ���2mL���ҵ�ú�ͣ��������¶�ճס����������ֽ���ɱ���ú�ͣ���ȥ�����㣩��
��1����ͼ��ʾ��װ��������һ֧С�ľ�֧�Թ��м���2mL���ҵ�ϡ���ᣬ�ټ���2mL���ҵ�ú�ͣ��������¶�ճס����������ֽ���ɱ���ú�ͣ���ȥ�����㣩��