��Ŀ����
9����֪���л���D��C4H7NO4����������嵰���ʵİ�����֮һ��������Aͨ�����·�Ӧ�ϳ���ȡ���л���A��C6H8O4��ΪʳƷ��װ�еij��÷�������������������ˮ������ʹ��ˮ��ɫ���л���B��C4H4O4�����������û��֧����ͨ��״����Ϊ��ɫ���壬��������������Һ������Ӧ����ش��������⣺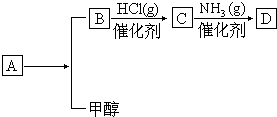
��1��B��D�Ľṹ��ʽ�ֱ���
 ��
��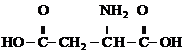 ��
����2��A���Է����ķ�Ӧ�Т٢ڢܣ�ѡ����ţ�
�ټӳɷ�Ӧ��������Ӧ��������Ӧ�ܼӾ۷�Ӧ
��3��B�����������Ĺ�����������̼̼˫�����Ȼ���
��4��B�ľ�����ͬ�����ŵ�ͬ���칹��Ľṹ��ʽ��
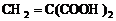 ��
����5��д����B��״���Ӧ��A�Ļ�ѧ����ʽ��HOOCCH=CHCOOH+2CH3OH$��_{��}^{Ũ����}$CH3OOCCH=CHCOOCH3+2H2O��
���� �л���D��C4H7NO4����������嵰���ʵİ�����֮һ�������Ȼ��Ͱ������л���A��C6H8O4������ʹ��ˮ��ɫ����̼̼˫�����л���B��C4H4O4�����������û��֧������������������Һ������Ӧ��BΪ��̼̼˫���Ķ�Ԫ���ᣬ����B�Ľṹ��ʽΪ
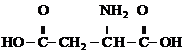
��� �⣺�л���D��C4H7NO4����������嵰���ʵİ�����֮һ�������Ȼ��Ͱ������л���A��C6H8O4������ʹ��ˮ��ɫ����̼̼˫�����л���B��C4H4O4�����������û��֧������������������Һ������Ӧ��BΪ��̼̼˫���Ķ�Ԫ���ᣬ����B�Ľṹ��ʽΪ
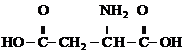
��1����������ķ�����֪��B�Ľṹ��ʽΪ
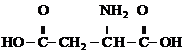
�ʴ�Ϊ��
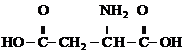
��2��AΪCH3OOCCH=CHCOOCH3��A���Է����ӳɷ�Ӧ��������Ӧ���Ӿ۷�Ӧ����ѡ�٢ڢܣ�
��3��B�Ľṹ��ʽΪ
�ʴ�Ϊ��̼̼˫�����Ȼ���
��4��B�Ľṹ��ʽΪ
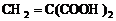
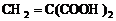
��5��B�Ľṹ��ʽΪ
�ʴ�Ϊ��HOOCCH=CHCOOH+2CH3OH$��_{��}^{Ũ����}$CH3OOCCH=CHCOOCH3+2H2O��
���� ���⿼���л�����ƶϣ��ؼ��Ǹ���A�����ʼ�B�ķ���ʽ���ṹ�ص�������ۺϷ���ȷ��B�Ľṹ��ʽ����Ŀ�Ѷ��еȣ�

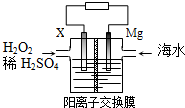
| A�� | X��Ϊ����ͭ��ʯī�ȵ缫 | |
| B�� | ÿת��2 mol���ӣ�2mol H+�ɽ���Ĥ������Ҳ�Ǩ�� | |
| C�� | �����ĵ缫��ӦʽΪH2O2+2e-+2H+�T2H2O | |
| D�� | �õ���ܽ���ѧ��ȫ��ת���ɵ��� |
| A�� | ���ڵ��Ȼ��� | B�� | CO2 | C�� | ʳ��ˮ | D�� | Fe |
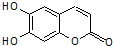 �в�ҩ��Ƥ�к��е���Ҷ�����������п������ã���1mol��Ҷ�������ֱ���Ũ��ˮ��NaOH��Һ��ȫ��Ӧ�������ĵ�Br2��NaOH�����ʵ����ֱ�Ϊ��������
�в�ҩ��Ƥ�к��е���Ҷ�����������п������ã���1mol��Ҷ�������ֱ���Ũ��ˮ��NaOH��Һ��ȫ��Ӧ�������ĵ�Br2��NaOH�����ʵ����ֱ�Ϊ��������| A�� | 2 mol Br2 2 mol NaOH | B�� | 2 mol Br2 3 mol NaOH | ||
| C�� | 3 mol Br2 4 mol NaOH | D�� | 4 mol Br2 4 mol NaOH |
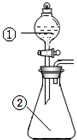 ��ͼ��ʾװ�ý�������ʵ�飺��������Һ������У�Ԥ���������ʵ��������ǣ�������
��ͼ��ʾװ�ý�������ʵ�飺��������Һ������У�Ԥ���������ʵ��������ǣ�������| ѡ�� | �������� | �������� | Ԥ����е����� |
| A | ϡ���� | ̼�������������ƵĻ����Һ | ������������ |
| B | Ũ���� | ��ɰֽ��ĥ�������� | ���������� |
| C | �Ȼ�����Һ | Ũ����������Һ | ����������ɫ���� |
| D | ϡ���� | ͭƬ | ����������������ɫ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | Ԫ��Y��Q���γɻ�����Y2O3 | |
| B�� | T��Z����һ�ֵ��ʵĿռ乹��Ϊ���������� | |
| C�� | X���������������幹��Ϊ������ | |
| D�� | ZO2�Ǽ��Լ����ɵķǼ��Է��ӣ�Zԭ�ӵ��ӻ���ʽΪsp�ӻ� |
| A�� | �����һ�ȼ��� | B�� | ���ۺ���ά�� | C�� | ���ͼױ� | D�� | ���Ǻ���ѿ�� |
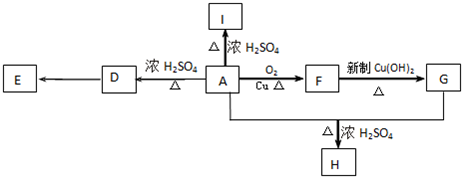
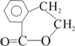 ��E
��E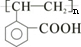 ��
��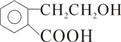 +O2
+O2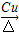 2
2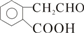 +2H2O��
+2H2O��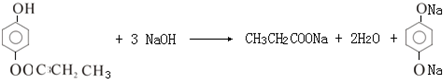 ��
��
 ��
��