��Ŀ����
��Ni��NiO��Ni3C����-Al2O3��ɵĴ������棬��ʵ��H2O��CO2��CH4�ϳ��Ҵ���
��1������˵����ȷ���� ��ѡ����ţ���
A����һ�����ܣ�̼ԭ�Ӵ�����ԭ��
B�� NO+����ʽΪ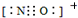
C��̼������CH3-��������
D�������ѵķе���Ҵ���
��2��Ni2+��̬�����Ų�ʽ ��
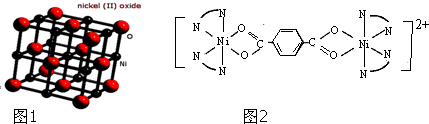
��3��NiO������ͼ1��ʾ��ÿ����������ÿ��Ni2+��������������O2-�� ��
��4���Ա������ᣨ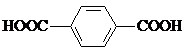 ����4��4��-�����
����4��4��-����� ���ɼ�Ϊ
���ɼ�Ϊ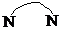 ������Ni2+���γɶ�����������[Ni2��TPHA����bpy��4]��ClO4��2�����ӵĽṹ��ͼ2��
������Ni2+���γɶ�����������[Ni2��TPHA����bpy��4]��ClO4��2�����ӵĽṹ��ͼ2��
TPHA���Ա������������� bpy��4-4�����
�����������λԭ���ǣ� ���Ա��������е�̼���ӻ���ʽΪ ��1mol 4��4��-����ऺ��ЦҼ�Ϊ ��
��1������˵����ȷ����
A����һ�����ܣ�̼ԭ�Ӵ�����ԭ��
B�� NO+����ʽΪ
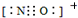
C��̼������CH3-��������
D�������ѵķе���Ҵ���
��2��Ni2+��̬�����Ų�ʽ
��3��NiO������ͼ1��ʾ��ÿ����������ÿ��Ni2+��������������O2-��
��4���Ա������ᣨ
 ����4��4��-�����
����4��4��-����� ���ɼ�Ϊ
���ɼ�Ϊ ������Ni2+���γɶ�����������[Ni2��TPHA����bpy��4]��ClO4��2�����ӵĽṹ��ͼ2��
������Ni2+���γɶ�����������[Ni2��TPHA����bpy��4]��ClO4��2�����ӵĽṹ��ͼ2��TPHA���Ա������������� bpy��4-4�����
�����������λԭ���ǣ�

���㣺Ԫ�������ɵ�����,ԭ�Ӻ�������Ų�,�����ijɼ����,�����ļ���,ԭ�ӹ���ӻ���ʽ���ӻ������ж�
ר�⣺
��������1��A�����ݵ�һ�����ܵĵݱ�����жϣ�
B��NO+�͵����������ڵȵ����壬�ȵ�����ṹ���ƣ�
C���ݼ۲���ӶԻ�������ȷ����ռ乹�ͣ�
D�����ݶ��ߵ������������жϣ�
��2������Niԭ�ӵ�ԭ������д����������Ų�ʽ��
��3�����ݾ����ṹ�ص�����жϣ�
��4�����������ӵĽṹ�ж���λԭ�ӣ����ݼ۲���ӶԻ�������ȷ�����ӻ���ʽ��4��4��-����ऺ��ЦҼ�����ĿΪ21��
B��NO+�͵����������ڵȵ����壬�ȵ�����ṹ���ƣ�
C���ݼ۲���ӶԻ�������ȷ����ռ乹�ͣ�
D�����ݶ��ߵ������������жϣ�
��2������Niԭ�ӵ�ԭ������д����������Ų�ʽ��
��3�����ݾ����ṹ�ص�����жϣ�
��4�����������ӵĽṹ�ж���λԭ�ӣ����ݼ۲���ӶԻ�������ȷ�����ӻ���ʽ��4��4��-����ऺ��ЦҼ�����ĿΪ21��
���
�⣺��1��A����һ������ͬ���ڴ����ң����������ƣ�Cλ��O����࣬��һ�����ܣ�̼ԭ��С����ԭ�ӣ���A����
B��NO+�͵����������ڵȵ����壬�ȵ�����ṹ���ƣ�����NO+����ʽΪ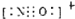 ����B��ȷ��
����B��ȷ��
C��̼������CH3-�۲���ӶԸ���=3+
��4+1-3��1��=4���Һ���һ���µ��Ӷԣ�����Ϊ�����Σ���C��ȷ��
D���Ҵ����Ӽ京����������Ҵ��۷е���ڶ����ѣ���D����
�ʴ�Ϊ��BC��
��2��Niԭ������Ϊ28�������������ԭ��������������ԭ���ͺ��ع���д��Ni2+��������Ų�ʽΪ[Ar]3d8��
�ʴ�Ϊ��[Ar]3d8��
��3���۲쾧����֪Ni2+λ�����ĺ����ģ�O2-λ�����ĺͶ��㣬��ÿ����������ÿ��Ni2+��������������O2-��6����
�ʴ�Ϊ��6��
��4��Nԭ���������5�����ӣ�ֻ���γ�3�����ۼ��ɴﵽ�ȶ��ṹ��������Ŀ��������Ϣ�����жϳ�����Ni2+�ɼ���Nԭ�Ӷ��γ���4�����ۼ����ɴ˿��ƶϳ�Ni2+�ṩ�չ����Nԭ���ṩ�¶Ե����γ���λ����ͬ�����Կ�����Ni2+���������O�γ���3�����ۼ�������֪�γ�����λ��������N��OΪ��λԭ�ӣ�
�Ա���������Cԭ�Ӷ�����3���Ҽ�����Cԭ�Ӳ���sp2�ӻ���
4��4��-����ऺ��ЦҼ�����ĿΪ21����1mol4��4��-����ऺ��ЦҼ�����ĿΪ21mol��
�ʴ�Ϊ��N��O��sp2�ӻ���21mol��
B��NO+�͵����������ڵȵ����壬�ȵ�����ṹ���ƣ�����NO+����ʽΪ
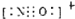 ����B��ȷ��
����B��ȷ��C��̼������CH3-�۲���ӶԸ���=3+
| 1 |
| 2 |
D���Ҵ����Ӽ京����������Ҵ��۷е���ڶ����ѣ���D����
�ʴ�Ϊ��BC��
��2��Niԭ������Ϊ28�������������ԭ��������������ԭ���ͺ��ع���д��Ni2+��������Ų�ʽΪ[Ar]3d8��
�ʴ�Ϊ��[Ar]3d8��
��3���۲쾧����֪Ni2+λ�����ĺ����ģ�O2-λ�����ĺͶ��㣬��ÿ����������ÿ��Ni2+��������������O2-��6����
�ʴ�Ϊ��6��
��4��Nԭ���������5�����ӣ�ֻ���γ�3�����ۼ��ɴﵽ�ȶ��ṹ��������Ŀ��������Ϣ�����жϳ�����Ni2+�ɼ���Nԭ�Ӷ��γ���4�����ۼ����ɴ˿��ƶϳ�Ni2+�ṩ�չ����Nԭ���ṩ�¶Ե����γ���λ����ͬ�����Կ�����Ni2+���������O�γ���3�����ۼ�������֪�γ�����λ��������N��OΪ��λԭ�ӣ�
�Ա���������Cԭ�Ӷ�����3���Ҽ�����Cԭ�Ӳ���sp2�ӻ���
4��4��-����ऺ��ЦҼ�����ĿΪ21����1mol4��4��-����ऺ��ЦҼ�����ĿΪ21mol��
�ʴ�Ϊ��N��O��sp2�ӻ���21mol��
���������⿼���Ϊȫ�棬�漰��һ�����ܡ��ȵ����塢����������Ų�ʽ�����ӿռ乹�͡��ӻ����͵��ж��Լ��йؾ���ļ��㣬�ѶȽϴ�ע������Ų�ʽ����д�ͷ��ӿռ乹�͵��жϣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
���и��������У�����ǿ������Һ�д���������ǣ�������
| A��Na+ K+ SO42- OH- |
| B��Na+ K+ Cl- HCO3- |
| C��Na+Cu2+ CO32- Br- |
| D��Na+ Mg2+ NO3- Cl- |
����˵������ȷ���ǣ�������
| A��������ˮ�����γ�Al��OH��3���壬��������ˮ�� |
| B�������뺣���������γ�ͨ���뽺��������й� |
| C���ü���ʼ��������Һ�Ķ����ЧӦ |
| D������е�1mol/L NaOH��Һ�еμ�FeCl3������Һ�Ʊ�Fe��OH��3���� |
��֪��Ӧ��2CO��g��+O2��g��?2CO2��g����H��0��ij�¶��£���2mol CO��1mol O2����10L�ܱ������У���Ӧ��ƽ���CO��ƽ��ת���ʣ���������ϵ��ѹǿ��p���Ĺ�ϵ��ͼ����ʾ��������˵����ȷ���ǣ�������


| A����ͼ��֪��A��CO��ƽ��Ũ��Ϊ0.4 mol?L-1 |
| B����ͼ��֪��B��CO��O2��CO2��ƽ��Ũ��֮��Ϊ2��l��2 |
| C����ƽ�����С�����ݻ�����Ӧ���ʱ仯ͼ�������ͼ�ұ�ʾ |
| D����0.50 MPaʱ��ͬ�¶���COת�������¶ȹ�ϵ���ͼ����T2��T1 |
�ܱ������У�������Ӧ��CO��g��+2H2��g��?CH3OH��g������H��0������˵����ȷ���ǣ�������
| A��һ���¶��£�ѹ�����������������Ӧ���ʼӿ죬�淴Ӧ���ʼ��� | ||
| B�����״����������ʵ���һ����̼���������ʣ���÷�Ӧ�ﵽƽ��״̬ | ||
C�������¶ȣ����´ﵽƽ��ʱ��
| ||
| D��ʹ�ô�������Ӧ��ƽ�ⳣ������ |
һ����A����B��Ӧ���ɷų�����ζ������C��C��NaOH��Һ��Ӧ����A��C����������D��D����ˮ������B����A��B�ֱ��ǣ�������
| A��Na2SO3��HNO3 |
| B��Na2SO3��H2SO4 |
| C��Na2S��H2SO4 |
| D��Na2S��HNO3 |
����˵����ȷ���ǣ�������
| A���ڼ�������Һ�зֱ���뱥��Na2SO4��CuSO4��Һ�������������������� |
| B��˾ĸ�춦����Զ���װ塢�л�������ԭ�������ںϽ� |
| C��������ά��̼��ά�����ά�������л��߷��Ӳ��� |
| D��ʯ�������ȷֽ�����˿���ʹ���Ը��������Һ��ɫ��ϩ�� |

