��Ŀ����
�±������ڱ��е�һ���֣�����A-I�����ڱ��е�λ�ã�����ĿҪ��ش�
��1������Ԫ�أ���ѧ��������õ��� ����������ǿ�ĵ����� �������ѧʽ��
��2������������ˮ����������ǿ���� ���ѧʽ����
��3��A�ֱ���D��E��F��G��H�γɵĻ������У����ȶ��� ���ѧʽ����
��4����B��C��E��F��G��H�У�ԭ�Ӱ뾶������ ����Ԫ�ط��ţ�
��5��A��D��ɻ�����ĵ���ʽ ��A��E��H��Ԫ����ɵĻ������������Ļ�ѧ��Ϊ ��
��6��B������������ˮ�����C��������������Ӧ�����ӷ���ʽ ��
| �� ���� | I A | ��A | ��A | ��A | ��A | ��A | ��A | O�� |
| 1 | A | |||||||
| 2 | D | E | G | I | ||||
| 3 | B | C | F | H |
��2������������ˮ����������ǿ����
��3��A�ֱ���D��E��F��G��H�γɵĻ������У����ȶ���
��4����B��C��E��F��G��H�У�ԭ�Ӱ뾶������
��5��A��D��ɻ�����ĵ���ʽ
��6��B������������ˮ�����C��������������Ӧ�����ӷ���ʽ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ÿ�֪��AΪ�⡢BΪNa��CΪMg��DΪ̼��EΪ����FΪ�ס�GΪ����HΪCl��IΪNe��
��1��ϡ�����廯ѧ��������ã��ǽ���Խǿ������������Խǿ��
��2������������ˮ����������ǿ���Ǹ����
��3���ǽ�����Խǿ���⻯��Խ�ȶ���
��4��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��5��A��D��ɵ��������ΪCH4��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl��
��6��B������������ˮ����ΪNaOH��C�����������ΪAl2O3�����߷�Ӧ����ƫ��������ˮ��
��1��ϡ�����廯ѧ��������ã��ǽ���Խǿ������������Խǿ��
��2������������ˮ����������ǿ���Ǹ����
��3���ǽ�����Խǿ���⻯��Խ�ȶ���
��4��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����
��5��A��D��ɵ��������ΪCH4��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl��
��6��B������������ˮ����ΪNaOH��C�����������ΪAl2O3�����߷�Ӧ����ƫ��������ˮ��
���
�⣺��Ԫ�������ڱ���λ�ÿ�֪��AΪ�⡢BΪNa��CΪMg��DΪ̼��EΪ����FΪ�ס�GΪ����HΪCl��IΪNe��
��1��ϡ������Ne�Ļ�ѧ��������ã�����Ԫ����F�ķǽ�����ǿ��F2��������ǿ���ʴ�Ϊ��Ne��F2��
��2������������ˮ����������ǿ���Ǹ����ᣬ��ѧʽΪHClO4���ʴ�Ϊ��HClO4��
��3��A�ֱ���D��E��F��G��H�γɵĻ�����Ϊ�⻯����з�Ԫ�طǽ�������ǿ����HF���ȶ����ʴ�Ϊ��HF��
��4��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶������B��C��E��F��G��H�У�ԭ�Ӱ뾶������Na���ʴ�Ϊ��Na��
��5��A��D��ɵ��������ΪCH4������ʽΪ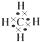 ��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl���������Ӽ������ۼ����ʴ�Ϊ��
��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl���������Ӽ������ۼ����ʴ�Ϊ��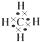 �����Ӽ������ۼ���
�����Ӽ������ۼ���
��6��B������������ˮ����ΪNaOH��C�����������ΪAl2O3�����߷�Ӧ����ƫ��������ˮ�����ӷ���ʽΪ��2OH?+Al2O3=2AlO2-+H2O��
�ʴ�Ϊ��2OH?+Al2O3=2AlO2-+H2O��
��1��ϡ������Ne�Ļ�ѧ��������ã�����Ԫ����F�ķǽ�����ǿ��F2��������ǿ���ʴ�Ϊ��Ne��F2��
��2������������ˮ����������ǿ���Ǹ����ᣬ��ѧʽΪHClO4���ʴ�Ϊ��HClO4��
��3��A�ֱ���D��E��F��G��H�γɵĻ�����Ϊ�⻯����з�Ԫ�طǽ�������ǿ����HF���ȶ����ʴ�Ϊ��HF��
��4��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶������B��C��E��F��G��H�У�ԭ�Ӱ뾶������Na���ʴ�Ϊ��Na��
��5��A��D��ɵ��������ΪCH4������ʽΪ
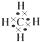 ��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl���������Ӽ������ۼ����ʴ�Ϊ��
��A��E��H��Ԫ����ɵĻ�����ΪNH4Cl���������Ӽ������ۼ����ʴ�Ϊ��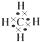 �����Ӽ������ۼ���
�����Ӽ������ۼ�����6��B������������ˮ����ΪNaOH��C�����������ΪAl2O3�����߷�Ӧ����ƫ��������ˮ�����ӷ���ʽΪ��2OH?+Al2O3=2AlO2-+H2O��
�ʴ�Ϊ��2OH?+Al2O3=2AlO2-+H2O��
���������⿼��Ԫ�����ڱ���Ԫ�������ɣ��ѶȲ���ע���Ԫ�����ڱ���������գ������ڻ���֪ʶ�Ĺ��̣�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
�����й�ͬ���칹����Ŀ�������У�����ȷ���ǣ�������
| A������ʽΪC5H10O2������8�� |
| B����Է�������Ϊ86������ͬ���칹����5�� |
| C���ױ�������һ����ԭ�ӱ���3��̼ԭ�ӵ����ȡ�������ò�����6�� |
| D������ʽΪC4H8Cl2���л�������к�2������ͬ���칹����4�֣����������칹�� |
��֪298K��101KPa������
2H2��g��+O2��g��=2H2O��g����H=-483.6kJ?mol-1
2H2��g��+O2��g��=2H2O��l����H=-571.6kJ?mol-1
�ݴ��жϣ�����˵����ȷ���ǣ�������
2H2��g��+O2��g��=2H2O��g����H=-483.6kJ?mol-1
2H2��g��+O2��g��=2H2O��l����H=-571.6kJ?mol-1
�ݴ��жϣ�����˵����ȷ���ǣ�������
| A��H2��g����O2��g����Ӧ����H2O��g�������ȷ�Ӧ |
| B��1molH2O��g��ת���1molH2O��l���ų�44.0kJ���� |
| C��1molH2O��l��ת���1molH2O��g���ų�44.0kJ���� |
| D��1molH2O��g��ת���1molH2O��l���ų�88.0kJ���� |
�������ֹ��ۼ����ӵ����ģ���У��ɱ�ʾCl2���ӵ��ǣ�������
A�� |
B�� |
C�� |
D�� |
NAΪ�����ӵ�����������������ȷ���ǣ�������
| A������£�2.8 L�����к��еļ��Թ��ۼ���ΪNA |
| B���ڷ�ӦKIO3+6HI=KI+3I2+3H2O �У�ÿ����3 mol I2ת�Ƶĵ�����Ϊ6 NA |
| C����֪CH3COONH4��Һ��PH=7����1L 0.1mol/L��CH3COONH4��Һ�У�CH3COO-��Ϊ0.1NA |
| D��39g Na2O2��Al��OH��3�Ļ���������������ӵ�����Ϊ2NA |
�����£�������CaCO3�������и���ϵ�У��ܽ�������ǣ�������
| A��10mLˮ |
| B��10mL 0.1mol/LCaCl2��Һ |
| C��20mL 0.1mol/LNaHSO4 |
| D��40mL 0.1mol/LNa2CO3��Һ |
���й����У����ڻ�ѧ�仯���ǣ�������
| A���ü��ȷ�������ɳ�еĵⵥ�� |
| B����������ȡ����ˮ |
| C���ɱ����� |
| D���������� |
���м��������е��ǣ�������
| A����ˮ�����Ҵ����ױ����屽 |
| B����ȼ�շ������Ҵ����������Ȼ�̼ |
| C�������Ը��������Һ���𱽡�����ϩ�ͻ����� |
| D����̼������Һ�����Ҵ���������������� |
 ���ݵ绯ѧ֪ʶ�ش��������⣮
���ݵ绯ѧ֪ʶ�ش��������⣮