��Ŀ����
5���ڵ�ˮ�����У���;��ҵ��ˮ�������ŷ������ˮ�ʶ�������������ij������Һ�к��д���Mg2+��Al3+��Cu2+��Ag+���Է����ش��������⣺��1���÷�Һ�п��ܴ������ڵ�һ����������B������ţ���
A��SO${\;}_{4}^{2-}$ B��NO${\;}_{3}^{-}$ C��Cl- D��CO${\;}_{3}^{2-}$
��2��Ϊ����Һ����Ԫ�صĺ����轫��ӷ�ˮ��Ʒ�з�����������õ��Լ���NaOH������þԪ�ط����仯�����ӷ���ʽ��Mg2++2OH-=Mg��OH��2����
��3��Ϊ�˻��շ�Һ�еĽ�������ijͬѧ�������ͼ������
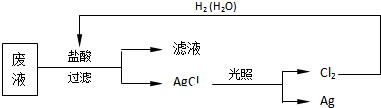
���÷������Ag 108g��Ϊ��֤����Ⱦ������Cl2��ѭ�����ã�������Ӧ�ṩ��״���µ�H2�������11.2L��
���� ��1��Ag+����������ӿɴ������棬���������������Ӿ�������ɳ������ܹ��棻
��2�����������Ӳ��ܴ����ڼ�����Һ�У����ˮ�����ԣ�����Һ����Ԫ�صĺ����轫��ӷ�ˮ��Ʒ�з��������Ӧ�ӹ����ļ���룻
��3���ɷ������̿�֪����Һ��ֻ��Ag+�����ᷴӦ���ɳ�����AgCl��������Ag�����2Ag��Cl2��H2���㣮
��� �⣺��1��Ag+����������ӿɴ������棬���������������Ӿ�������ɳ������ܹ��棬���ɵ���غ㼰���ӹ����֪����Һ��������һ��ΪB��
�ʴ�Ϊ��B��
��2�����������Ӳ��ܴ����ڼ�����Һ�У����ˮ�����ԣ�����Һ����Ԫ�صĺ����轫��ӷ�ˮ��Ʒ�з��������Ӧ�ӹ����ļ���룬���Լ�ΪNaOH��þԪ�ط����仯�����ӷ���ʽ�� Mg2++2OH-=Mg�� OH��2����
�ʴ�Ϊ��NaOH��Mg2++2OH-=Mg�� OH��2����
��3���ɷ������̿�֪����Һ��ֻ��Ag+�����ᷴӦ���ɳ�����AgCl��������Ag������ڹ�ϵʽ2Ag��Cl2��H2��������Ӧ�ṩ��״���µ�H2�������$\frac{108g}{108g/mol}$��$\frac{1}{2}$��22.4L/mol=11.2L���ʴ�Ϊ��11.2��
���� ���⿼����������ᴿ�����ӹ��桢���ӷ�Ӧ��Ϊ��Ƶ���㣬��������֮��ķ�Ӧ�����ʵ�������ؼ���Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע���غ㷨Ӧ�ã���Ŀ�ѶȲ���

C��s��+O2��g���TCO2��g�� H=-393.51KJ/mol
��ӦC ��s��+CO2��g���T2CO��g���ġ�HΪ��������
| A�� | -283.01KJ/mol | B�� | +172.51 KJ/mol | C�� | +283.01KJ/mol | D�� | -504.01 KJ/mol |
| A�� | 1mol•L-1 | B�� | 2mol•L-1 | C�� | 3mol•L-1 | D�� | 4mol•L-1 |
| A�� | Ư��¶���ڿ����У�ClO-+CO2+H2O�THClO+HCO3- | |
| B�� | ��������ˮ���ն�������SO2+NH3•H2O�THSO3-+NH4+ | |
| C�� | ����CO2ͨ�뱽������Һ��2C6H5O-+CO2+H2O��2C6H5OH+CO32- | |
| D�� | ������������Fe3O4����������Fe3O4+8H+�TFe2++2Fe3++4H2O |
���ݢ١�⑫Ԫ�������ڱ��е�λ�ã��ش������й����⣺�����û�ѧ������д��
| IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 | |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� | �� | |
| 4 | �� | ⑪ | ⑫ |
��2������ЩԪ�ص�����������Ӧˮ�����У�������ǿ����HClO4�������Ե�����������Al��OH��3�����û�ѧʽ��д��
��3������ЩԪ���е��ʵ���������ǿ����F2����ԭ����ǿ����K����ѧ��������õ�Ԫ����Ar�����û�ѧʽ��д��
��4���õ���ʽ��ʾ�ߺ͢��γɻ�����Ĺ��̣�
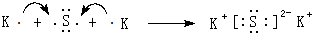 ��
����5���ߺ͢�ֱ��γɵ���̬�⻯������Բ��ȶ�����H2S��
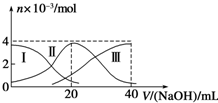
| A�� | ��Ũ�ȵ�NaOH��Һ��H2A��Һ��2��1��Ϻ�����Һ��ˮ�ĵ���̶ȱȴ�ˮ�� | |
| B�� | ��V��NaOH��=20 mLʱ����Һ������Ũ�ȴ�С��ϵ��c��Na+����c��HA-����c��A2-����c��H+����c��OH-�� | |
| C�� | NaHA��Һ�У�c��OH-��=c��H+��+c��HA-��+2c��H2A�� | |
| D�� | ����������20mLNaOH��Һ��������Һ���ټ���ˮ�Ĺ����У�pH���ܼ��� |
| A�� | �ֱ���H2O2��KMnO4�ֽ���O2�����Ƶõ�����O2ʱ��ת�Ƶ�����֮��Ϊ1��2 | |
| B�� | ��������O2��O3�У���ԭ�ӵĸ�����Ϊ3��2 | |
| C�� | CaCl2��CaO2������������������֮�Ⱦ�Ϊ2��1 | |
| D�� | 10mL 0.3mol/LNaCl��30mL0.1mol/LAlCl3��Һ��Cl-���ʵ���Ũ�ȱ�Ϊ1��3 |

 �����������д��ڵĻ�ѧ���й��ۼ���
�����������д��ڵĻ�ѧ���й��ۼ��� ��
��