��Ŀ����
A��B��CΪ������Ԫ�أ�AΪ�����Ľ�����B�ڳ�����Ϊ���壬��������������ˮ����DΪ����ǿ�ᣬC������������ˮ����EΪ����ǿ�A��B��C�����������ת����ϵ��ͼ��
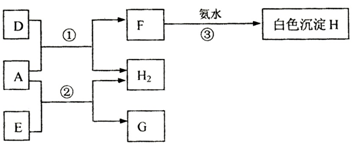
��ش��������⣺
��1��д��D�Ļ�ѧʽ______��B��C�γɵĻ�����ĵ���ʽΪ______��
��2��д����Ӧ�۵����ӷ���ʽ______��
��3����״���£���������A�ֱ���������D��E��Ӧ������H2������ֱ�ΪV1��V2�����ϵΪV1______V2���������=������V2=2.24l����Ӧ������ת�Ƶĵ��ӵ����ʵ���Ϊ______��
�⣺A��B��CΪ������Ԫ�أ�AΪ�����Ľ�����B�ڳ�����Ϊ���壬��������������ˮ����DΪ����ǿ���ж�ΪH2SO4��C������������ˮ����EΪ����ǿ��ͽ���A��Ӧ�����������ƶ�EΪNaOH��A�ȿ��Ժ�ǿ�ᷴӦҲ���Ժ�ǿ�Ӧ�ж�ΪAl��A��D��H2SO4����Ӧ���ɵ�FΪ��������HΪAl��OH��3��GΪNaAlO2����������ƶϵõ�BΪ��Ԫ�أ�CΪ��Ԫ�أ�
��1��D�Ļ�ѧʽΪ��H2SO4��B��C�γɵĻ�����ΪNa2S�ĵ���ʽΪ��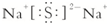
�ʴ�Ϊ��H2SO4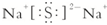 ��
��
��2����Ӧ�����������Ͱ�ˮ��Ӧ�����������������Ĺ��̣���Ӧ�����ӷ���ʽΪ��Al3++3NH3?H2O=Al��OH��3��+3NH4+���ʴ�Ϊ��Al3++3NH3?H2O=Al��OH��3��+3NH4+��
��3����״���£���������A��Al���ֱ���������D��H2SO4����E��NaOH����Ӧ������H2������ֱ�ΪV1��V2�����ݵ����غ�õ��������������ͬ����V2=2.24l��n��H2��=0.1mol��Ӧ������ת�Ƶĵ��ӵ����ʵ���Ϊ0.2mol��
�ʴ�Ϊ��0.2mol��
������AΪ�����Ľ�����B�ڳ�����Ϊ���壬��������������ˮ����DΪ����ǿ���ж�ΪH2SO4��C������������ˮ����EΪ����ǿ��ͽ���A��Ӧ�����������ƶ�EΪNaOH��A�ȿ��Ժ�ǿ�ᷴӦҲ���Ժ�ǿ�Ӧ�ж�ΪAl��A��D��H2SO4����Ӧ���ɵ�FΪ��������HΪAl��OH��3��GΪNaAlO2����������ƶϵõ�BΪ��Ԫ�أ�CΪ��Ԫ�أ������жϽ��з����ش�
���������⿼��������ת����ϵ��Ӧ�ã��������ʵ�Ӧ�ã���Ҫ������ǿ��������ʵ�Ӧ�á���Ӧ���ļ��㣬����ʽ����д�����ӷ���ʽ����д�������жϣ�
��1��D�Ļ�ѧʽΪ��H2SO4��B��C�γɵĻ�����ΪNa2S�ĵ���ʽΪ��
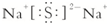
�ʴ�Ϊ��H2SO4
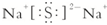 ��
����2����Ӧ�����������Ͱ�ˮ��Ӧ�����������������Ĺ��̣���Ӧ�����ӷ���ʽΪ��Al3++3NH3?H2O=Al��OH��3��+3NH4+���ʴ�Ϊ��Al3++3NH3?H2O=Al��OH��3��+3NH4+��
��3����״���£���������A��Al���ֱ���������D��H2SO4����E��NaOH����Ӧ������H2������ֱ�ΪV1��V2�����ݵ����غ�õ��������������ͬ����V2=2.24l��n��H2��=0.1mol��Ӧ������ת�Ƶĵ��ӵ����ʵ���Ϊ0.2mol��
�ʴ�Ϊ��0.2mol��
������AΪ�����Ľ�����B�ڳ�����Ϊ���壬��������������ˮ����DΪ����ǿ���ж�ΪH2SO4��C������������ˮ����EΪ����ǿ��ͽ���A��Ӧ�����������ƶ�EΪNaOH��A�ȿ��Ժ�ǿ�ᷴӦҲ���Ժ�ǿ�Ӧ�ж�ΪAl��A��D��H2SO4����Ӧ���ɵ�FΪ��������HΪAl��OH��3��GΪNaAlO2����������ƶϵõ�BΪ��Ԫ�أ�CΪ��Ԫ�أ������жϽ��з����ش�
���������⿼��������ת����ϵ��Ӧ�ã��������ʵ�Ӧ�ã���Ҫ������ǿ��������ʵ�Ӧ�á���Ӧ���ļ��㣬����ʽ����д�����ӷ���ʽ����д�������жϣ�

��ϰ��ϵ�д�
 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�
�����Ŀ
|
A��B��CΪ������Ԫ�أ����������ڱ��е�λ������ͼ��ʾ����֪B��C��Ԫ���������ڱ�������֮����AԪ��������2����B��CԪ�ص�ԭ������֮����AԪ��ԭ��������4������A��B��C�ֱ�Ϊ
| |
A�� |
Be��Na��Al |
B�� |
B��Mg��Si |
C�� |
C��Al��P |
D�� |
O��P��Cl |
A��B��CΪ������Ԫ�أ����������ڱ��е�λ������ͼ��ʾ����֪B��C��Ԫ���������ڱ�������֮����AԪ��������2����B��CԪ�ص�ԭ������֮����AԪ��ԭ��������4������A��B��C�ֱ�Ϊ
|
|
A |
|
|
B |
|
C |
A��Be Na Al B��B Mg Si C��C Al P D��O P Cl
A��B��CΪ������Ԫ�أ����������ڱ��е�λ������ͼ��ʾ����֪B��C��Ԫ���������ڱ�������֮����AԪ��������2����B��CԪ�ص�ԭ������֮����AԪ��ԭ��������4������A��B��C�ֱ�Ϊ
| A | ||
| B | C |
- A.Be Na Al
- B.B Mg Si
- C.C Al P
- D.O P Cl

