��Ŀ����
2009�꡶��Ȼ����־�������ҹ���ѧ��ͨ������SiO2��26Al��10Be����Ԫ�صı���ȷ���������ˡ�������о���������ֲ��������С�������귨���� �����������գ�
��1��10Be��9Be ��������ţ�
a��������ͬ�Ļ�ѧ����
b��Be Ԫ�صĽ������ԭ��������9.5
c��������ͬ��������
d��ͨ����ѧ�仯����ʵ��10Be��9Be����ת��
��2���о�����26Al����˥��Ϊ26Mg�����ԱȽ�������Ԫ�ؽ�����ǿ���ķ����� ��������ţ�
a���Ƚ�������Ԫ�صĵ��ʵ�Ӳ�Ⱥ��۵�
b���������з����Ѿõ�������Ԫ�صĵ��ʷֱ����ˮ����
c������ĥ����þ������Ƭ�ֱ����ˮ���ã��������̪��Һ
d�����Ȼ������Ȼ�þ����Һ�зֱ�μӹ���������������Һ
��3��Ŀǰ����һ�ֲ��������С���벲��귨����д����Ar��������Ų���ͬ�������ӵİ뾶�ɴ�С��˳�� ���û�ѧ���ű�ʾ��������һ�������������Ԫ�ص��������γɵĻ�����������ɲټ����˻�����ĵ���ʽΪ ��
��1��10Be��9Be
a��������ͬ�Ļ�ѧ����
b��Be Ԫ�صĽ������ԭ��������9.5
c��������ͬ��������
d��ͨ����ѧ�仯����ʵ��10Be��9Be����ת��
��2���о�����26Al����˥��Ϊ26Mg�����ԱȽ�������Ԫ�ؽ�����ǿ���ķ�����
a���Ƚ�������Ԫ�صĵ��ʵ�Ӳ�Ⱥ��۵�
b���������з����Ѿõ�������Ԫ�صĵ��ʷֱ����ˮ����
c������ĥ����þ������Ƭ�ֱ����ˮ���ã��������̪��Һ
d�����Ȼ������Ȼ�þ����Һ�зֱ�μӹ���������������Һ
��3��Ŀǰ����һ�ֲ��������С���벲��귨����д����Ar��������Ų���ͬ�������ӵİ뾶�ɴ�С��˳��
���㣺ͬһ������Ԫ�����ʵĵݱ������ԭ�ӽṹ�Ĺ�ϵ,ͬλ�ؼ���Ӧ��,���뾶��С�ıȽ�
ר�⣺
��������1��10Be��9Be����������ͬ����������ͬ������ͬλ�أ�
��2�������Ľ�����ǿ���ıȽϷ����У�����֮����û���Ӧ��������ͬ�����ˮ�������������׳̶ȡ�����������������ˮ�������ǿ�������жϣ�
��3����Ar��������Ų���ͬ����������S2-��Cl-�����Ӳ�ṹ��ͬ�����ӣ������Ӱ뾶����ԭ���������������С������һ�������������Ԫ�ص��������γɵĻ������������������ø������CaCl2��CaCl2����������֮��������Ӽ���
��2�������Ľ�����ǿ���ıȽϷ����У�����֮����û���Ӧ��������ͬ�����ˮ�������������׳̶ȡ�����������������ˮ�������ǿ�������жϣ�
��3����Ar��������Ų���ͬ����������S2-��Cl-�����Ӳ�ṹ��ͬ�����ӣ������Ӱ뾶����ԭ���������������С������һ�������������Ԫ�ص��������γɵĻ������������������ø������CaCl2��CaCl2����������֮��������Ӽ���
���
�⣺��1��a��10Be��9Be����ͬ��Ԫ�ص�ԭ�ӣ���ѧ������ͬ����a��ȷ��
b��Be Ԫ�صĽ������ԭ������������ͬλ�ص��������ĺ͵�һ�룬�����ͬλ�صķ���йأ��ʴ���
c��10Be��9Be���������ֱ�Ϊ6��5����������������ͬ���ʴ���
d����ѧ�仯��ԭ�ӱ��ֲ��䣬��ͨ����ѧ�仯����ʵ��10Be��9Be����ת�����ʴ���
�ʴ�Ϊ��a��
��2��a��������ǿ�����䵥�ʵ�Ӳ�Ⱥ��۵��أ��ʴ���
b���������з����Ѿõ�������Ԫ�صĵ��ʷֱ����ˮ���ã����߱��涼���������ﱡĤ������þ��������������ˮ��Ӧ���������жϽ�����ǿ�����ʴ���
c������ĥ����þ������Ƭ�ֱ����ˮ���ã��������̪��Һ��Mg�ͷ�ˮ����Һ�ʺ�ɫ��Al�ͷ�ˮ����Һ����ɫ��˵��Mg���û�����������Mg�Ļ����Դ���Al������ȷ��
d�����Ȼ������Ȼ�þ����Һ�зֱ�μӹ���������������Һ����������������NaOH��Һ����������þ������NaOH��Һ������Mg�Ľ����Դ���Al������ȷ��
�ʴ�Ϊ��cd��
��3����Ar��������Ų���ͬ����������S2-��Cl-�����Ӳ�ṹ��ͬ�����ӣ������Ӱ뾶����ԭ���������������С���������������Ӱ뾶��С˳����S2-��Cl-������һ�������������Ԫ�ص��������γɵĻ������������������������Ǹ����ӣ��ø������CaCl2��CaCl2����������֮��������Ӽ��������ʽΪ ��
��
�ʴ�Ϊ��S2-��Cl-�� ��
��
b��Be Ԫ�صĽ������ԭ������������ͬλ�ص��������ĺ͵�һ�룬�����ͬλ�صķ���йأ��ʴ���
c��10Be��9Be���������ֱ�Ϊ6��5����������������ͬ���ʴ���
d����ѧ�仯��ԭ�ӱ��ֲ��䣬��ͨ����ѧ�仯����ʵ��10Be��9Be����ת�����ʴ���
�ʴ�Ϊ��a��
��2��a��������ǿ�����䵥�ʵ�Ӳ�Ⱥ��۵��أ��ʴ���
b���������з����Ѿõ�������Ԫ�صĵ��ʷֱ����ˮ���ã����߱��涼���������ﱡĤ������þ��������������ˮ��Ӧ���������жϽ�����ǿ�����ʴ���
c������ĥ����þ������Ƭ�ֱ����ˮ���ã��������̪��Һ��Mg�ͷ�ˮ����Һ�ʺ�ɫ��Al�ͷ�ˮ����Һ����ɫ��˵��Mg���û�����������Mg�Ļ����Դ���Al������ȷ��
d�����Ȼ������Ȼ�þ����Һ�зֱ�μӹ���������������Һ����������������NaOH��Һ����������þ������NaOH��Һ������Mg�Ľ����Դ���Al������ȷ��
�ʴ�Ϊ��cd��
��3����Ar��������Ų���ͬ����������S2-��Cl-�����Ӳ�ṹ��ͬ�����ӣ������Ӱ뾶����ԭ���������������С���������������Ӱ뾶��С˳����S2-��Cl-������һ�������������Ԫ�ص��������γɵĻ������������������������Ǹ����ӣ��ø������CaCl2��CaCl2����������֮��������Ӽ��������ʽΪ
 ��
���ʴ�Ϊ��S2-��Cl-��
 ��
��
���������⿼����ۺϣ��漰ͬλ�ء����Ӱ뾶��С�Ƚϡ�������ǿ���жϡ�����ʽ��֪ʶ�㣬��Щ֪ʶ�㶼�ǿ��Ը�Ƶ�㣬���ݻ������Ԫ�������ɵ�֪ʶ�������������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 �ŵ������ϵ�д�
�ŵ������ϵ�д� 53������ϵ�д�
53������ϵ�д�
�����Ŀ
�ס��ҡ��������ֱ���Al2��SO4��3��FeSO4��NaOH��BaCl2���������е�һ�֣���������Һ��������Һ�У������а�ɫ�������ɣ������μ��������ʧ������Һ�������Һʱ�����������������ݴ˿��жϱ������ǣ�������
| A��Al2��SO4��3 |
| B��NaOH |
| C��BaCl2 |
| D��FeSO4 |
�����з�Ӧ�У�������������ܺ��ڷ�Ӧ��������ܺ͵��ǣ�������
| A��Zn��s��+CuSO4��aq��=ZnSO4��aq��+Cu��s�����ȷ�Ӧ |
| B��2CO��g��+O2��g��=2CO2��g�����ȷ�Ӧ |
| C��CaCO3��s��=CaO��s��+CO2��g�����ȷ�Ӧ |
| D��H+��aq��+OH-��aq��=H2O��l�����ȷ�Ӧ |
���й�����Χ�����Ų�ʽΪ3s23p4������������ȷ���ǣ�������
| A������ԭ�Ӻ�����������״��ͬ�ĵ����� |
| B������ԭ�Ӻ�����ӹ���16�ֲ�ͬ���˶�״̬ |
| C������Ԫ�ط���ΪO |
D��������ӹ����ʾʽΪ |
�п�Ժ��������ѧ����2013��11��22�������������Ŀ�����Ա�ڹ������״Ρ��ġ�������ġ���Ƭ����ʵ���������ʵ�ռ����Ϊ������ı��ʡ���һ��ѧ��������80����������ṩ��ֱ��֤�ݣ��ⲻ������������������ʶ��ǰ�ƽ���һ��ҲΪ�ڷ��ӡ�ԭ�ӳ߶��ϵ��о��ṩ�˸���ȷ�ķ���������˵������ȷ���ǣ�������
| A����������Ĵ��ڣ����ܸ���ˮ���� |
| B���������Ȼ��������Ҫ��������㷺�Ļ�ѧ��֮һ |
| C����������Ĵ��ڣ��е㣺HCl��HBr��HI��HF |
| D����������Ĵ��ڣ�ˮ���ӱ�ĸ��ȶ��� |
��3.84��ͭ����һ������Ũ���ᷴӦ����ͭ��ȫ����ʱ���ռ�������4.48L������£�������������������ʵ����ǣ�������
| A��0.26mol |
| B��0.32mol |
| C��0.16mol |
| D��0.2mol |
�������У���ʹˮ�ĵ���ƽ�������ƶ�����ˮ��Һ�ʼ��Ե��ǣ�������
A��aX+�� |
B�� |
| C��X2-����Ar������ͬ�����Ų� |
| D��26M3+ |
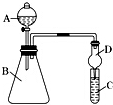 ������ͼװ�ÿ�����֤�ǽ����Եı仯���ɣ�
������ͼװ�ÿ�����֤�ǽ����Եı仯���ɣ�
 ��ͼ��������֮������ߴ�����ѧ�����絥����˫��������A�Ľṹ��ʽΪ
��ͼ��������֮������ߴ�����ѧ�����絥����˫��������A�Ľṹ��ʽΪ