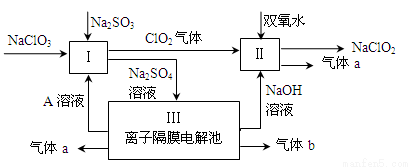
��Ŀ����
ijѧ����NaHCO3��KHCO3��ɵ�ij�����������ʵ�飬������±����ݣ���������ʵ���Ũ����ȣ���
ʵ���� | �� | �� | �� |
������� | 50mL���� | 50mL���� | 50mL���� |
m(�����) | 9.2g | 16.56g | 27.6g |
V(CO2)(��״��) | 2.24L | 3.36L | 3.36L |
�����������ݣ�����˵������ȷ����( )
A���ɢٿ��Լ������������ʵ���Ũ��
B���ɢڡ��ۿ�֪��������������ӣ��������û�б仯��˵�������Ѿ���Ӧ��ȫ
C���ɢ١��ڿ�֪�����еĹ��岻�����ȫ��Ӧ
D���ɢٿ��Լ����������NaHCO3����������
A
��������
�����������������Ӧ��H++HCO3-�TCO2��+H2O���ɢ٢����ݿ�֪���������������������������������˵��ʵ�����������ʣ�ࡢ����������ȫ��Ӧ��������������ʣ�࣬����������������������������������ɢڢ����ݿ�֪��ʵ���������������䣬˵��ʵ�����������ȫ��Ӧ������ʵ��ٵ����ݿ�֪������3.36L������̼��Ҫ�������������Ϊ9.2g��2.24L��3.36L=13.8g��16.56g����ʵ�����������ȫ��Ӧ������������ʣ�ࡣA��������������֪��ʵ�����������ʣ�࣬���ܸ���ʵ��ټ������������ʵ���Ũ�ȣ�ʵ�����������ȫ��Ӧ��ʵ����ж�����̼�����ݿ��Լ�����������ʵ���Ũ�ȣ���A����B��������������ʣ�࣬����������������������������������ɢڢ����ݿ�֪��ʵ���������������䣬˵��ʵ�����������ȫ��Ӧ����B��ȷ��C���ɢ٢����ݿ�֪���������������������������������˵��ʵ�����������ʣ�ࡢ����������ȫ��Ӧ����C��ȷ��D������9.2g�����ʱ��������ʣ�࣬�������ȫ��Ӧ������H++HCO3-�TCO2��+H2O����֪n������=n��CO2��=0.1mol����9.2 g�������NaHCO3��KHCO3�����ʵ����ֱ�Ϊx��y�����У�x+y=0.1mol��84x+100y=9.2g���������̼��㣬����������Ե��������ʿ��Լ���NaHCO3��������������D��ȷ��
���㣺���⿼������ļ��㡣

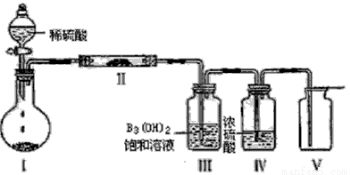
 ��У����ϵ�д�
��У����ϵ�д���10�֣��±��г������ڱ���������6��Ԫ�ص��й����ݣ�
Ԫ�ر�� | �� | �� | �� | �� | �� | �� |
ԭ�Ӱ뾶��nm�� | 0.075 | 0.110 | 0.152 | 0.071 | 0.186 | 0.099 |
���̬ | +5 | +5 | +1 | 0 | +1 | +7 |
��ͼ�̬ | ��3 | ��3 | 0 | ��1 | 0 | ��1 |
�����ϱ���Ϣ��գ�
��1��д�����б�Ŷ�ӦԪ�ص�Ԫ�ط��ţ��� ���� ��
��2��Ԫ�آ������ڱ��е�λ���� ��
��3��Ԫ�آ��γɵ����ֳ���������Ļ�ѧʽ�ֱ�Ϊ �����ж����еĻ�ѧ���� ��
��4���ٵ��⻯��͢��⻯�ﷴӦ�Ļ�ѧ����ʽΪ ��
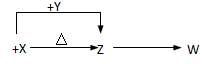
 H2O(g) + CO(g)����Ӧ�������¶���ͬ������ʼŨ�� ��ͬ������ �ף�n(CO2) �� n(H2) �� 1 mol�� �ң�n(CO2)��1 mol�� n(H2)�� 2 mol������n(CO2) �� n(H2)��n��H2O(g)�ݣ�1 mol.�ﵽƽ��ʱCO�����ʵ����ɴ�С��˳����
H2O(g) + CO(g)����Ӧ�������¶���ͬ������ʼŨ�� ��ͬ������ �ף�n(CO2) �� n(H2) �� 1 mol�� �ң�n(CO2)��1 mol�� n(H2)�� 2 mol������n(CO2) �� n(H2)��n��H2O(g)�ݣ�1 mol.�ﵽƽ��ʱCO�����ʵ����ɴ�С��˳����