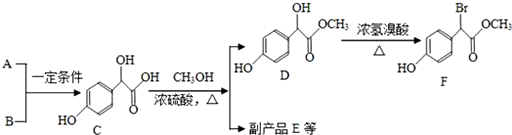
��Ŀ����
18��ij�л�������A�����ϣ��������к�̼Ϊ72.0%������Ϊ6.67%������Ϊ�����������з����ⶨ���л����������Է��������ͷ��ӽṹ������һ��������������֪A����Է�������Ϊ150��
���������˴Ź����Dz��A�ĺ˴Ź���������5���壬�����֮��Ϊ1��2��2��2��3��
�����������ú�������Dz��A���ӵĺ��������ͼ��ʾ��
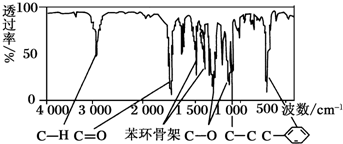
����գ�
��1��A�ķ���ʽΪC9H10O2��
��2��A�ķ����к�һ������������bc��
a��A����Է������� b��A�ķ���ʽ
c��A�ĺ˴Ź�������ͼ d��A���ӵĺ������ͼ
��3��A�Ľṹ��ʽΪC6H5COOCH2CH3��C6H5CH2COOCH3��C6H5OOCCH2CH3��
��4��A�ķ�����ͬ���칹���ж��֣����з��������������ٷ��ӽṹ��ֻ��һ�������ţ��ڷ��ӽṹ�к���һ�������۱�����ֻ��һ��ȡ�����������A��ͬ���칹�干��6�֣�����A����
���� ��1���л���A�����У�N��C��=$\frac{150��72%}{12}$=9��N��H��=$\frac{150��6.67%}{1}$=10����N��O��=$\frac{150-12��9-10}{16}$=2�������л���A�ķ���ʽΪC9H10O2��
��2��A�ĺ˴Ź���������5���壬˵����������5��Hԭ�ӣ������֮��Ϊ��Ӧ�ĸ���Hԭ�Ӹ���֮�ȣ����ݷ���ʽ��֪������Hԭ������������ȷ������Ŀ��
��3����A���ӵĺ������֪������C6H5C-���ţ��ɱ�����ֻ��һ��ȡ������֪�������ϵ��������֣�Hԭ�Ӹ����ֱ�Ϊ1����2����2������A�ĺ˴Ź�������֪���������⣬���������⣬��������ĸ����ֱ�Ϊ2����3������A���ӵĺ������֪��A���ӽṹ��̼̼������C-H��C=O��C-O�Ȼ��ţ�
��4������Ŀ������֪��ȡ���������Ϊ-C3H5O2������������ȡ�����У�-OOCCH2CH3��-COOCH2CH3��-CH2OOCCH3��-CH2COOCH3��-CH��CH3��OOCH��-CH��CH3��COOH��
��� �⣺��1���л���A�����У�N��C��=$\frac{150��72%}{12}$=9��N��H��=$\frac{150��6.67%}{1}$=10����N��O��=$\frac{150-12��9-10}{16}$=2�������л���A�ķ���ʽΪC9H10O2��
�ʴ�Ϊ��C9H10O2��
��2��A�ĺ˴Ź���������5���壬˵����������5��Hԭ�ӣ������֮��Ϊ��Ӧ�ĸ���Hԭ�Ӹ���֮�ȣ��ɷ���ʽ��֪������Hԭ������������ȷ������Ŀ��������Ҫ֪��A�ķ���ʽ��A�ĺ˴Ź�������ͼ��
�ʴ�Ϊ��bc��
��3����A���ӵĺ������֪������C6H5C-���ţ��ɱ�����ֻ��һ��ȡ������֪�������ϵ��������֣�Hԭ�Ӹ����ֱ�Ϊ1����2����2������A�ĺ˴Ź�������֪���������⣬���������⣬��������ĸ����ֱ�Ϊ2����3������A���ӵĺ������֪��A���ӽṹ��̼̼�������������ţ����Է����������л���A�ṹ��ʽΪC6H5COOCH2CH3��C6H5CH2COOCH3��C6H5OOCCH2CH3��
�ʴ�Ϊ��C6H5COOCH2CH3��C6H5CH2COOCH3��C6H5OOCCH2CH3��
��4������Ŀ������֪��ȡ���������Ϊ-C3H5O2������������ȡ�����У�-OOCCH2CH3��-COOCH2CH3��-CH2OOCCH3��-CH2COOCH3��-CH��CH3��OOCH��-CH��CH3��COOH��
�ʴ�Ϊ��6��
���� ���⿼���л�����ƶϡ��л������ʽȷ������������ͬ���칹����д�ȣ�������ѧ���ķ��������ͼ��������Ŀ��飬ע������л���Ľṹ�����ʵĹ�ϵ��������ͼд�����������Ŀ��ܽṹ��ʽ��

| A�� | 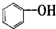 ���ڴ������ ���ڴ������ | B�� |  ���ڷ����廯���� ���ڷ����廯���� | ||
| C�� |  ����֬�������� ����֬�������� | D�� | CH3CH��CH3��2������״������ |
| A�� | 1 mol O2��Ϊ�������õ��ĵ�����Ϊ2NA | |
| B�� | ��0�棬101kPaʱ��22.4 L�����к���NA����ԭ�� | |
| C�� | NA��һ����̼���Ӻ�0.5 mol�����������Ϊ7��4 | |
| D�� | 25�棬1.01��105Pa��64gSO2�к��е�ԭ����Ϊ3NA |

�����й�˵������ȷ���ǣ�������
| A�� | ������ת���Ƕ���������ͼ�е��ȼҵ��һ��������ת��Ϊ��ѧ�ܵĹ��� | |
| B�� | ���̢��нᾧ����MgCl2•6H2OҪ��HCl��Χ�м�����ˮ�Ƶ���ˮMgCl2 | |
| C�� | ���̢��г�ȥ�����е�SO42-��Ca2+��Mg2+��Fe3+�����ʣ����뻯ѧҩƷ��˳��Ϊ��Na2CO3��Һ��NaOH��Һ��BaCl2��Һ�����˺������ | |
| D�� | �ڹ��̢ۢ�����Ԫ�ؾ������� |
 ��
��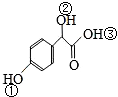 �Т١��ڡ���3��-OH��������ǿ������˳���ǣ��ۣ��٣��ڣ�
�Т١��ڡ���3��-OH��������ǿ������˳���ǣ��ۣ��٣��ڣ�
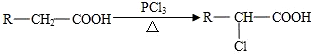
 ��
�� ��Br2��Ҫ��õ������IJ������NaOH��Һ����ˮ���Լ�ϴ�ӣ�ϴ�Ӻ����ֲ�ƷӦʹ�õ������Ƿ�Һ©����
��Br2��Ҫ��õ������IJ������NaOH��Һ����ˮ���Լ�ϴ�ӣ�ϴ�Ӻ����ֲ�ƷӦʹ�õ������Ƿ�Һ©���� ��
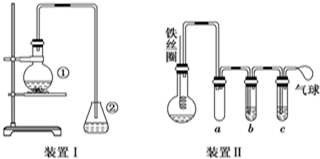
��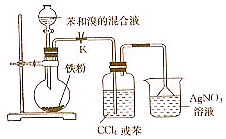 ʵ��������ͼ��ʾװ�ý��б����巴Ӧ��ʵ�飬��ش��������⣮
ʵ��������ͼ��ʾװ�ý��б����巴Ӧ��ʵ�飬��ش��������⣮ +Br2$\stackrel{FeBr_{3}}{��}$
+Br2$\stackrel{FeBr_{3}}{��}$ +HBr��
+HBr��