��Ŀ����
14���������ǵ��������ı����������������и�ǿ�������ԣ�ʵ���ҿɽ�����ͨ����ѹ�ŵ������ȡ������3O2$\frac{\underline{\;�ŵ�\;}}{\;}$2O3����1������������Ӧ����30%������ת��Ϊ���������û������ƽ��Ħ������Ϊ35.6 g•mol-1������һλС������
��2����8L����ͨ���ŵ�ָܺ���ԭ״�����õ�����6.5L�����г���Ϊ3 L��
��3��ʵ���ҽ������ͳ����Ļ������0.896L����״����ͨ��ʢ��20.0gͭ�۵ķ�Ӧ���У���ּ��Ⱥ�ĩ��������Ϊ21.6g����ԭ������г������������Ϊ50%��
��4�������£���ȡ��ͬ����������Ʒ����ˮ���������к���pH=7��Ȼ����Һ���ɵ��Ȼ��ƾ��壬���ɹ����в�Ʒ����ʧ��
| ��������������g�� | �Ȼ���������g�� | |
| �� | 2.40 | 3.51 |
| �� | 2.32 | 2.34 |
| �� | 3.48 | 3.51 |
���� ��1���ȸ���ת���ʣ����÷���ʽ��������Ե����ʵ�����Ȼ�����ƽ��Ħ����������������������������ʵ���֮�ȣ���Ӧ������У����������������䣬���ʵ�����С��
��2������������������ò������������ɳ����������
��3������0.896L����������ʵ���Ϊ$\frac{0.896L}{22.4L/mol}$=0.04mol����̬ȫ�����뷴Ӧ�������������������������������ʵ����з��̼���������ʵ���������������������������
��4������n=$\frac{m}{M}$�������ʵ����NaCl�����ʵ���������ȷ���������Ƶ����ʵ������ٸ���M=$\frac{m}{n}$�����������������Ʒ��Ħ���������ݴ�ȷ���Ƿ��нᾧˮ��ȷ����Ʒ��ɽ����ó����ۣ�
��� �⣺��1��������1molO2����Ӧ��ת����Ϊ30%��������Ӧ��O2Ϊ��1mol��30%=0.3mol��
3O2$\frac{\underline{\;�ŵ�\;}}{\;}$2O3
��ʼ 1mol 0
��Ӧ 0.3mol 0.2mol
ƽ�� 0.7mol 0.2mol
�ʷ�Ӧ����������Ϊ0.2molO3��0.7molO2��ƽ��Ħ������ΪM=$\frac{0.7mol��32g/mol+0.2mol��48g/mol}{0.7mol+0.2mol}$=35.6 g/mol��
�ʴ�Ϊ��35.6��
��2��3O2$\frac{\underline{\;�ŵ�\;}}{\;}$2O3 ��V
2 1
V��O3�� ��8-6.5��L=1.5L
��ã�V��O3��=2��1.5 L=3L��
�ʴ�Ϊ��3��
��3��0.896L����������ʵ���Ϊ$\frac{0.896L}{22.4L/mol}$=0.04mol����Ӧ�������������21.6g-20g=1.6g����̬ȫ�����뷴Ӧ�������������������ʵ����ֱ�Ϊxmol��ymol�����ݶ����������������ʵ�������$\left\{\begin{array}{l}{x+y=0.04}\\{32x+48y=1.6}\end{array}\right.$
��ã�x=0.02��y=0.02���ʳ������������Ϊ$\frac{0.02mol}{0.04mol}$��100%=50%��
�ʴ�Ϊ��50%��
��4��ʵ�����NaCl�����ʵ���=$\frac{3.51g}{58.5g/mol}$=0.06mol�����������غ㣬��NaOH��Ʒ�����ʵ���Ϊ0.06mol������������Ʒ��Ħ������=$\frac{2.4g}{0.06mol}$=40g/mol����Ϊ��ȡ����������ƷΪNaOH��
ʵ�����NaCl�����ʵ���=$\frac{2.34g}{58.5g/mol}$=0.04mol�����������غ㣬��NaOH��Ʒ�����ʵ���Ϊ0.04mol������������Ʒ��Ħ������=$\frac{2.32g}{0.04mol}$=58g/mol����Ϊ��ȡ����������Ʒ���нᾧˮ���ᾧˮ��Ŀ=$\frac{58-40}{18}$=1������ȡ����������ƷӦ��NaOH��H2O��
ʵ�����NaCl�����ʵ���=$\frac{3.51g}{58.5g/mol}$=0.06mol�����������غ㣬��NaOH��Ʒ�����ʵ���Ϊ0.06mol������������Ʒ��Ħ������=$\frac{3.48g}{0.06mol}$=58g/mol����Ϊ��ȡ����������Ʒ���нᾧˮ���ᾧˮ��Ŀ=$\frac{58-40}{18}$=1������ȡ����������ƷӦ��NaOH��H2O��
�������������ɵó����ۣ�ʵ�����ȡ����������ƷΪNaOH��ʵ��ڢ���ȡ����������ƷΪNaOH��H2O��
�𣺿ɵó����ۣ�ʵ�����ȡ����������ƷΪNaOH��ʵ��ڢ���ȡ����������ƷΪNaOH��H2O��
���� ���⿼��������ɵ�ȷ�����ؼ���ȷ������������Ʒ�нᾧˮ���⣬���ؿ���ѧ����ʵ�����ݷ��������������Ѷ��еȣ�

 �ƸԹھ��ο���ϵ�д�
�ƸԹھ��ο���ϵ�д�| A�� | HCl�ĵ���ʽ 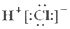 | B�� | Cl-�Ľṹʾ��ͼ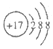 | ||
| C�� | CO2�ĵ���ʽ 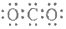 | D�� | ������Ϊ37����ԭ�� ${\;}_{37}^{17}$Cl |
��1�������ͬѧ���Ʊ�ԭ����NO��Cl2���Ʊ�װ����ͼ1��ʾ��
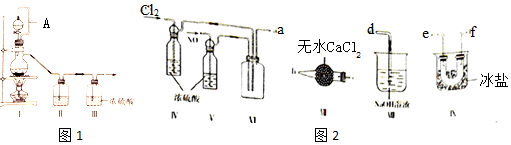
Ϊ�Ʊ�������������壬����ȱ�ٵ�ҩƷ�ǣ�
| װ�â� | װ�â� | ||
| ��ƿ�� | ��Һ©���� | ||
| �Ʊ�����Cl2 | MnO2 | �� | �� |
| �Ʊ�����NO | Cu | �� | �� |
��װ������˳��Ϊa��e��f����f��e����c��b��d ���������������ҷ�����Сд��ĸ��ʾ����
��װ�� IV��V ���ɽ�һ������NO��Cl2�⣬��һ�������ǹ۲����ݵ�����������٣�
��װ�� VII�������Ƿ�ֹˮ�������뷴Ӧ����
��װ�� VIII������β��ʱ��NOCl������Ӧ�Ļ�ѧ����ʽΪNOCl+2NaOH=NaCl+NaNO2+H2O��
��3������ͬѧ�������ϣ������ˮ��Ũ������Ũ����Ļ��ᣬһ�������»������������̪�Ⱥ��������÷�Ӧ�Ļ�ѧ����ʽΪHNO3��Ũ��+3HC1��Ũ��=NOCl��+Cl2��+2H2O��
| A�� | �������������ᷴӦ�����κ�ˮ�����Թ��������Ǽ��������� | |
| B�� | ij��������ɫ��ӦΪ��ɫ����������Ϊ�������� | |
| C�� | Na2O2���������Ӹ�����1��l | |
| D�� | ���������������̼��Ӧʱ���������Ƽ������������ǻ�ԭ�� |
| A�� | NaOH | B�� | KMnO4 | C�� | KSCN | D�� | ����KI |
| A�� | AgNO3��Һ | B�� | KI������ֽ | ||
| C�� | ϡHNO3��AgNO3��Һ | D�� | NaBr��Һ��CCl4 |
| A�� | H2CO3?H2O+CO2�� | B�� | KOH�TK++O2-+H+ | ||
| C�� | HClO�TH++ClO- | D�� | Na2CO3�T2Na++CO32- |