��Ŀ����
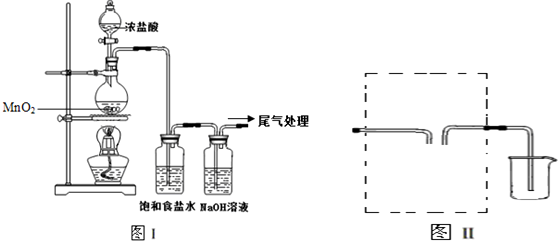
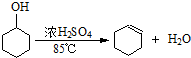
7��ij��ѧС�����ͼ1װ�ã��Ի������Ʊ�����ϩ��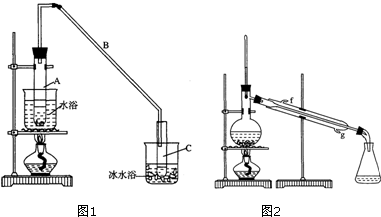
��֪��
| �ܶȣ�g/cm3�� | �۵㣨�棩 | �е㣨�棩 | �ܽ��� | |
| ������ | 0.96 | 25 | 161 | ������ˮ |
| ����ϩ | 0.81 | -103 | 83 | ������ˮ |
��1���Ʊ���Ʒ
��12.5mL�����������Թ�A�У��ټ���1mLŨ���ᣬҡ�Ⱥ�������Ƭ��������������Ӧ��ȫ�����Թ�C�ڵõ�����ϩ��Ʒ��
�ٵ���B���˵�������е�������������
�ڼ����Թ�A������ˮԡ������ֱ�Ӽ��ȣ�Ŀ���ǿ����¶�85�����ң���ֹ�������ӷ���ʹ���Ⱦ��ȣ��Թ�C���ڱ�ˮԡ�е�Ŀ������������ϩ����ֹ��ӷ���
��2���Ʊ���Ʒ
�ٻ���ϩ��Ʒ�к��л������������������ʵȣ����뱥��ʳ��ˮ�������á��ֲ㣬����ϩ���� �㣨��ϡ����¡�������Һ����C�������ţ�ϴ�ӣ�
A��KMnO4��Һ B��ϡH2SO4 C��Na2CO3��Һ
���ٽ�����ϩ��ͼ2װ��������ȴˮ��g�ڽ��루����ĸ����
���ռ���Ʒʱ�����Ƶ��¶�Ӧ��83�����ң�ʵ���ƵõĻ���ϩ��Ʒ�����������۲��������ܵ�ԭ����C��
A������ʱ��70�濪ʼ�ռ���Ʒ
B��������ʵ����������
C���Ʊ���Ʒʱ���������Ʒһ������
��3���ƻ���ϩʱ������Ũ�����ǿ�����ԣ�����������SO2��CO2��ˮ��������С���������Լ�����
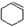 ��SO2��CO2��ˮ�������������ͨ���Լ���˳���Ǣܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ� ������ţ�
��SO2��CO2��ˮ�������������ͨ���Լ���˳���Ǣܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ� ������ţ��ٺ�Na2SO3��Һ ������KMnO4��Һ ��ʯ��ˮ ����ˮCuSO4 ��Ʒ����Һ��
���� ��1���ٵ���B���˵���������������ã����ڻ���ϩ������
����ˮԡ���ȿ��Կ����¶�85�����ң���ֹ�������ӷ�����ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ����
��2���ٻ���ϩ�������Ȼ�����Һ�����ܶȱ�ˮС���ֲ��ϩ���ϲ㣬���ڷ�Һ��ϩ��Ʒ�л�������������ͻ��������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�
��Ϊ����������Ч������ȴˮ���¿ڣ�g�����룬��ȴˮ�������γ�����������Ч�����ã������������ܳ���ˮ���Է�������ڴ���������ʹ���������ѣ�
�۸��ݱ������ݿ�֪����ֻ���ϩ�ķе�Ϊ83�棻
a������ǰ�ռ�����Ʒ�л������ʣ�ʵ�ʲ����������۲�����
b����ȡ�Ļ���ϩ���ʵ�������ʵ���ƵõĻ���ϩ��Ʒ�����������۲�����
c���ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�����
��3������ ����������KMnO4��Һ������ˮ����ͭ����ˮ����Ʒ�����������������Ը�����س�ȥ�������������ʯ��ˮ���������̼��
����������KMnO4��Һ������ˮ����ͭ����ˮ����Ʒ�����������������Ը�����س�ȥ�������������ʯ��ˮ���������̼��
��� �⣺��1���ٵ���B���˵���������������ã����ڻ���ϩ������
�ʴ�Ϊ��������
����ˮԡ���ȿ��Կ����¶�85�����ң���ֹ�������ӷ���ʹ���Ⱦ��ȣ���ˮԡ��Ŀ���ǽ��ͻ���ϩ�������¶ȣ�ʹ��Һ����
�ʴ�Ϊ�������¶�85�����ң���ֹ�������ӷ���ʹ���Ⱦ��ȣ���������ϩ����ֹ��ӷ���
��2���ٻ���ϩ�����࣬�������Ȼ�����Һ�����ܶȱ�ˮС�������á��ֲ��ϩ���ϲ㣬���ڷ�Һ��ϩ��Ʒ�л�������������ͻ����������룺�Ʊ����������ᴿ����ʱ��c��Na2CO3��Һ��ϴ�ӿɳ�ȥ�ᣬ
�ʴ�Ϊ���ϲ㣻c��
��Ϊ����������Ч��������װ��Ҫ�������ܣ���ȴˮ���¿ڣ�g�����룬��ȴˮ�������γ�����������Ч�����ã�
�ʴ�Ϊ��g��
�۸��ݱ������ݿ�֪����ֻ���ϩ�ķе�Ϊ83�棬���ռ���ƷӦ�����¶���83�����ң�
a������ʱ��70�濪ʼ�ռ���Ʒ����ǰ�ռ�����Ʒ�л������ʣ�ʵ�ʲ����������۲�������a����
b��������ʵ���������ˣ���ȡ�Ļ���ϩ�����ʵ�������ʵ���ƵõĻ���ϩ��Ʒ�����������۲�������b����
c�����ֲ�Ʒ�л��л����������²ⶨ���ĵĻ������������ƵõĻ���ϩ��Ʒ�����������۲�������c��ȷ������ѡc��
�ʴ�Ϊ��83�棻C��
��3������ ����������KMnO4��Һ������SO2����������KMnO4��Һ��ɫ��Ʒ����Һ��ʯ��ˮ������CO2����ʯ��ˮ������ˮ����������ˮCuSO4�������ڼ���������������迼���Լ���ѡ���˳��ֻҪͨ����Һ���ͻ����ˮ����������ȼ���ˮ������Ȼ�����SO2���ڼ���֮���ȥSO2����SO2�����ñ���Na2SO3��Һ��������CO2�ͱ�ϩ�����˳��Ϊ�ܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ���
����������KMnO4��Һ������SO2����������KMnO4��Һ��ɫ��Ʒ����Һ��ʯ��ˮ������CO2����ʯ��ˮ������ˮ����������ˮCuSO4�������ڼ���������������迼���Լ���ѡ���˳��ֻҪͨ����Һ���ͻ����ˮ����������ȼ���ˮ������Ȼ�����SO2���ڼ���֮���ȥSO2����SO2�����ñ���Na2SO3��Һ��������CO2�ͱ�ϩ�����˳��Ϊ�ܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ���
�ʴ�Ϊ���ܢݢ٢ݢڢۣ���ܢݢ٢ݢۢڣ���
���� ���⿼�����Ի������Ʊ�����ϩ��ʵ�鷽������Ŀ�Ѷ��еȣ��ۺ��Խ�ǿ������ʱע��������ʵķ��롢�ᴿ�������������ʵ����ʵ���ͬ�ǽ�����Ĺؼ������ؿ���ѧ�����ʵ�������������

 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д�| A�� | ���ô�����ˮ | B�� | ϴθ | ||
| C�� | ������ҩ�� | D�� | ͣҩ������ע��NaHCO3��Һ |
| A�� | NaOH��Һ | B�� | ��ˮ | C�� | ���� | D�� | Na2SiO3��Һ |
| A�� | �������ڼ��� | B�� | Ư�����ڻ���� | ||
| C�� | ��������������� | D�� | ������̼�������������� |
| A�� | ��Һ����ȡ������ | B�� | ��ȡ����Һ������ | C�� | ��ȡ������Һ | D�� | ��Һ��������ȡ |

 ��þ�����Ļ���ﹲ0.2mol������200mL 4mol•L-1��������Һ�У�Ȼ���ٵμ�2mol•L-1��NaOH��Һ��
��þ�����Ļ���ﹲ0.2mol������200mL 4mol•L-1��������Һ�У�Ȼ���ٵμ�2mol•L-1��NaOH��Һ��