��Ŀ����
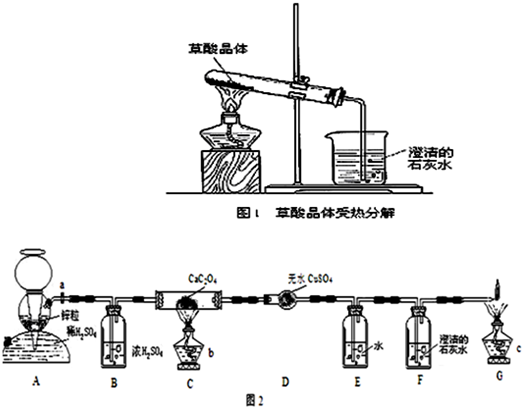
�̿��飺����ͼ1��ʾ�����Թ��е��Ҷ��ᾧ�壨H2C2O4?2H2O�����ȣ����ֽ������ͨ�����ġ����С�ձ��У�Ȼ����ͨ������ʯ��ˮ�У��۲�����
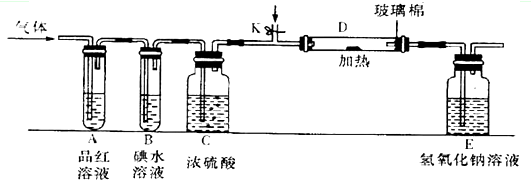
���ڷ���ʵʩ�����У��������±ˣ����Թܿ�������б���Ҷ��ᾧ���ۻ���Һ�壬˳�Թܱ������Թܿڣ�����ʵ�鲻�ܳɹ����ڽ��ֽ����ͨ�����ġ����С�ձ��У����������ˮ�������Ҷ��ᾧ��ֽ�����Ľᾧˮ������֤���Ҷ���ֽ��ˣ����Ҷ������Ȼӷ�������ͨ������ʯ��ˮ�У����ɵ�CaC2O4�ǰ�ɫ����������֤CO2�и��ţ���ȷ֤�Ҷ���ֽ⣻���Ҷ�����ȷֽ�IJ������ΪCO2��CO��H2O������CO���ж����壬���봦�����ȣ�
������������ij�о���ѧϰС�����̽������Ƴ�����ʵ������Ҷ���ķֽⲢ�ⶨ��ֽ��ʣ������������£�
�ٰ��Ҷ��ᾧ����ں����н��к濾��ȥ���ᾧˮ�����ã�
�ڰ�ͼ2���Ӻ�װ�ã�
�ۼ��װ�õ������ԣ�
�ܴ���a��ͨ��H2һ������ٹر�a����ȼ�ƾ���b��c��
�ݵ�Cװ���й�����ʧ��ֹͣ����
����a������ͨ��H2��ֱ����ȴ��
�Իش��������⣺
��1��װ��B������ ��װ��E������ ��
��2���������װ�������Եķ����� ��
��3��������װ��A��B����ʹ�����Ҷ���ķֽ��� �����������С�����䡱����
��4�����Դ���װ��G�ķ����� ��
��5������ȡH2C2O4����ɣ�������Ϊ4.5g��ʵ������Ƶ�D��E��F�ֱ�����0.95g��0.40g��1.98g�����Ҷ���ķֽ���Ϊ ��

���ڷ���ʵʩ�����У��������±ˣ����Թܿ�������б���Ҷ��ᾧ���ۻ���Һ�壬˳�Թܱ������Թܿڣ�����ʵ�鲻�ܳɹ����ڽ��ֽ����ͨ�����ġ����С�ձ��У����������ˮ�������Ҷ��ᾧ��ֽ�����Ľᾧˮ������֤���Ҷ���ֽ��ˣ����Ҷ������Ȼӷ�������ͨ������ʯ��ˮ�У����ɵ�CaC2O4�ǰ�ɫ����������֤CO2�и��ţ���ȷ֤�Ҷ���ֽ⣻���Ҷ�����ȷֽ�IJ������ΪCO2��CO��H2O������CO���ж����壬���봦�����ȣ�
������������ij�о���ѧϰС�����̽������Ƴ�����ʵ������Ҷ���ķֽⲢ�ⶨ��ֽ��ʣ������������£�
�ٰ��Ҷ��ᾧ����ں����н��к濾��ȥ���ᾧˮ�����ã�
�ڰ�ͼ2���Ӻ�װ�ã�
�ۼ��װ�õ������ԣ�
�ܴ���a��ͨ��H2һ������ٹر�a����ȼ�ƾ���b��c��
�ݵ�Cװ���й�����ʧ��ֹͣ����
����a������ͨ��H2��ֱ����ȴ��
�Իش��������⣺
��1��װ��B������
��2���������װ�������Եķ�����
��3��������װ��A��B����ʹ�����Ҷ���ķֽ���
��4�����Դ���װ��G�ķ�����
��5������ȡH2C2O4����ɣ�������Ϊ4.5g��ʵ������Ƶ�D��E��F�ֱ�����0.95g��0.40g��1.98g�����Ҷ���ķֽ���Ϊ
���㣺����ʵ�鷽�������
ר�⣺ʵ�������
������ʵ�����̵�ԭ���Ǹ����Ҷ������ȷֽ����������Լ�������������ȷ���ֽ�IJ��Ϊ��֤������ﱻ������գ���ʼ��ȡ�����������������������崵���������װ���У���������ȡ�к���ˮ������������Ũ�����������ó����ʯ��ˮ�����������̼�IJ�����������װ�õ���������ȷ�������Ķ�����̼������Ϊ��ֹ�Ҷ���Ļӷ����Ž����������ˮ�����Ҷ��ᣬһ����̼���ж������壬�����������ռ���
��1��Ũ���������ˮ�ԣ�������������Ҷ���������ˮ�����ʣ�
��2�������շ������Ļ���a���۲�B��E��Fƿ���Ƿ������ݲ������������������õģ�
��3��ʵ��ɰܵĹؼ����Ҷ���ֽ����������ȫ�������գ�װ���в�����ʣ��ķֽ����ݴ˻ش�
��4��CO�ж�������������У�����ȼ���⣬���������ң������ռ�������
��5�����������������ɵ�CO2���������H2C2O4
CO2��+CO��+H2O���õ��ֽ�IJ�����������ݴ˼���ֽ��ʣ�
��1��Ũ���������ˮ�ԣ�������������Ҷ���������ˮ�����ʣ�
��2�������շ������Ļ���a���۲�B��E��Fƿ���Ƿ������ݲ������������������õģ�
��3��ʵ��ɰܵĹؼ����Ҷ���ֽ����������ȫ�������գ�װ���в�����ʣ��ķֽ����ݴ˻ش�
��4��CO�ж�������������У�����ȼ���⣬���������ң������ռ�������
��5�����������������ɵ�CO2���������H2C2O4
| ||
���
�⣺ʵ�����̵�ԭ���Ǹ����Ҷ������ȷֽ����������Լ�������������ȷ���ֽ�IJ��Ϊ��֤������ﱻ������գ���ʼ��ȡ�����������������������崵���������װ���У���������ȡ�к���ˮ������������Ũ�����������ó����ʯ��ˮ�����������̼�IJ�����������װ�õ���������ȷ�������Ķ�����̼������Ϊ��ֹ�Ҷ���Ļӷ����Ž����������ˮ�����Ҷ��ᣬһ����̼���ж������壬�����������ռ���
��1��������շ������Ƶõ������к���ˮ�����������ȥ��װ��B�������Ǹ���H2�����ȥ�����л��е�ˮ���������Ҷ���Ϊ�л�����Ȼ�ӷ�������������Ӧ��ͨ��ˮ�м����ܽ⣬�Ա���Ժ���ʵ��������ţ��ʴ�Ϊ������H2�����ȥ�����л��е�ˮ���������ܽ�ӷ������Ҷ����������������ʵ�飻
��2�������װ��ʹ�õ������϶࣬�Ҷദ�е���ͨ��Һ���У��ɴ����շ������Ļ���a������������ͨ��B��E��Fƿ�������ݲ������ɼ����������ԣ��ʴ�Ϊ�������շ������Ļ���a���۲�B��E��Fƿ���Ƿ������ݲ������������ݲ�����˵����©�������������ã�
��3����ͨ��H2��Ŀ����ʹ�Ҷ���ֽ����������ȫ��ͨ��D��E��Fװ�ã����������ֽ����������������װ��C�У�����װ��A��B����D��E��Fװ�����ӵ���������С�����õ��ķֽ��ʼ��٣�
�ʴ�Ϊ����С��
��4����������õ�����ΪCO���ж�������������У�����ȼ���⣬���������ң������ռ�������
�ʴ�Ϊ�������ң������ռ�������
��5����H2C2O4
CO2��+CO��+H2O������F����1.98g�������ɵ�CO2����Ϊ1.98g���ɵó��ֽ��H2C2O4Ϊ4.05g�����Ҷ���ķֽ���Ϊ
��100%=90%����������У����ܲ���D��Eװ�õ��������ݣ���δ�ֽ���Ҷ�������Dװ������ȴ��Ҳ����˵0.95g����ȫ������ˮ��������0.40g������ȫ��δ�ֽ���Ҷ����������
�ʴ�Ϊ��90%��
��1��������շ������Ƶõ������к���ˮ�����������ȥ��װ��B�������Ǹ���H2�����ȥ�����л��е�ˮ���������Ҷ���Ϊ�л�����Ȼ�ӷ�������������Ӧ��ͨ��ˮ�м����ܽ⣬�Ա���Ժ���ʵ��������ţ��ʴ�Ϊ������H2�����ȥ�����л��е�ˮ���������ܽ�ӷ������Ҷ����������������ʵ�飻
��2�������װ��ʹ�õ������϶࣬�Ҷദ�е���ͨ��Һ���У��ɴ����շ������Ļ���a������������ͨ��B��E��Fƿ�������ݲ������ɼ����������ԣ��ʴ�Ϊ�������շ������Ļ���a���۲�B��E��Fƿ���Ƿ������ݲ������������ݲ�����˵����©�������������ã�
��3����ͨ��H2��Ŀ����ʹ�Ҷ���ֽ����������ȫ��ͨ��D��E��Fװ�ã����������ֽ����������������װ��C�У�����װ��A��B����D��E��Fװ�����ӵ���������С�����õ��ķֽ��ʼ��٣�
�ʴ�Ϊ����С��
��4����������õ�����ΪCO���ж�������������У�����ȼ���⣬���������ң������ռ�������
�ʴ�Ϊ�������ң������ռ�������
��5����H2C2O4
| ||
| 4.05g |
| 4.5g |
�ʴ�Ϊ��90%��
����������Ŀ��һ������ʵ�鷽��������⣬����ѧ�����ʵ������Լ�ʵ�鷽������ƣ�����ѧ�������ͽ��������������Ѷȴ�

��ϰ��ϵ�д�
�����Ŀ
�����й�˵����ȷ���ǣ�������
| A����NH3ͨ���ȵ�CuSO4��Һ����ʹCu2+��ԭ��Cu |
| B�������£�������ˮ���ܴ�������C1-��Fe3+��NO3-��Na+��SO32- |
| C��Ũ�Ⱥ������ͬ��NaOH��CH3COOH���ϣ���Һ��c��Na+����c��CH3COO-�� |
| D���ŵ練ӦΪH2+2NiO��OH��=2Ni��OH��2�ĵ�أ����ʱ��������NiO��OH������ԭ |
��֪298Kʱ���ϳɰ���ӦN2��g��+3H2��g���T2NH3��g����H=-92.0kJ?mol-1�������¶��µ�1mol N2��3mol H2����һ�ܱ������У��ڴ�������ʱ���з�Ӧ����÷�Ӧ�ų�������Ϊ���ٶ�����������û��������ʧ����������
| A��һ������92.0 kJ |
| B����ȷ�� |
| C��һ��С��92.0 kJ/mol |
| D��һ��С��92.0 kJ |
�����½�������Һ�������Ϻ�������Һ��pHһ������7���ǣ�������
| A��pH=4�������pH=10�İ�ˮ |
| B��pH=4�Ĵ�����Һ��pH=10������������Һ |
| C��0.1mol?L-1�������0.1mol?L-1������������Һ |
| D��0.1mol?L-1�������0.1mol?L-1������������Һ |
�����й�ԭ�ӽṹ��Ԫ�������ɵ���������ȷ���ǣ�������
| A��ԭ�Ӱ뾶��Na��O�����Ӱ뾶��Na+��O2- |
| B���ڢ�A��Ԫ�ص�����ֺ���131I��137I�ĺ��������֮��Ϊ6 |
| C��P��As���ڵ�VA��Ԫ�أ�H3PO4������ǿ��H3AsO4������ |
| D��Na��Mg��Al����Ԫ������������Ӧˮ����ļ��Ծ����μ��� |
�ڽ�����������У�����һ��ʱ��������Ƭ���ӣ�Ӧ��ȡ����ȷ�����ǣ�������
| A���������� | B����ȴ�� |
| C�����貹�� | D���������� |