��Ŀ����
4��������ȾԽ��Խ��Ϊ���ǹ�ע�����⣬�����е�NOx�����ѳ�����������������ŷţ������ķ����ж��֣����������գ�
��ֱ������
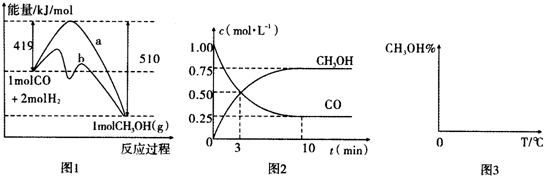
��1��NO�ڴ��������·ֽ�Ϊ��������������10L�ܱ������У�NO��ֱ��������Ӧʱ�������ʵ����仯��ͼ1��ʾ����0��5min��������ƽ����Ӧ����Ϊ0.015mol/��L•min����

���������
��2��O3����NO ���ˮϴ����ȫת��ΪHNO3����ʱO3��NO�����ʵ���֮��Ϊ1��2��
��������
��3��ʵ������ȡ�����İ����������Ȼ���⣬����Ҫ�죨����ʯ�ҡ���ʯ�ң���д�Լ����ƣ�����ʹ��̼��淋�ԭ���ǣ�NH4��2CO3$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+CO2��+2H2O���û�ѧ����ʽ��ʾ����
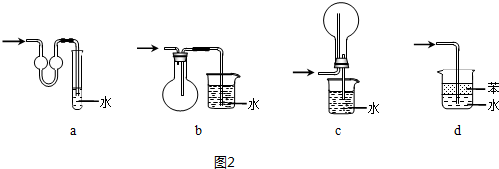
��4�����հ���ʱ����ʹ�÷�����װ�ã�ͼ2װ�ò��ܴﵽ��Ŀ����d��
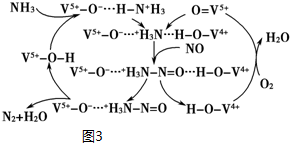
NH3�ѳ�������NO��ԭ����ͼ3��
��5��������ԭ���У�NO����ת��ΪN2���ѧʽ����H2O��������1mol NH3��0.25mol O2ʱ����ȥ��NO�ڱ�״���µ����Ϊ22.4L��
���� ��1��ͼ��õ�NO���ʵ����仯������õ��������ʵ����仯����ϻ�ѧ��Ӧ���ʸ������V=$\frac{��c}{��t}$��
��2��O3����NO ���ˮϴ����ȫת��ΪHNO3����Ӧ�Ļ�ѧ����ʽΪ��O3+2NO+H2O=2HNO3��
��3��ʵ�������ù����Ȼ�狀��������Ƽ��ȷ�Ӧ���ɣ������Ǽ������壬������Ҫ���Ը��������ʹ��̼��淋�ԭ����̼��識������ɰ���ͬʱ�����ɶ�����̼���壻
��4��������������ˮ����ˮ��Һ��������������������ѹǿ��֪ʶ���з�����
��5������ͼ3��֪��Ӧ��Ϊ������һ�������Ͱ�������������Ϊ������ˮ��
�ڸ��ݰ���ʧȥ�ĵ��ӵ����ʵ�������NO�������õ��ĵ��������ʵ������㣮
��� �⣺��1����10L�ܱ������У�NO��ֱ��������Ӧʱ�������ʵ����仯��ͼ1��ʾ��NO��5min�ڱ仯���ʵ���8.0mol-6.5mol=1.5mol��NO�ڴ��������·ֽ�Ϊ������������2NO=O2+N2�������������ʵ���0.75mol����0��5min��������ƽ����Ӧ����=$\frac{\frac{0.75mol}{10L}}{5min}$=0.015mol/L•min��
�ʴ�Ϊ��0.015��
��2��O3����NO ���ˮϴ����ȫת��ΪHNO3����Ӧ�Ļ�ѧ����ʽΪ��O3+2NO+H2O=2HNO3����ʱO3��NO�����ʵ���֮��Ϊ1��2��
�ʴ�Ϊ��1��2��
��3��ʵ�������ù����Ȼ�狀��������Ƽ��ȷ�Ӧ���ɣ������Ǽ������壬������Ҫ���Ը������ʵ������ȡ�����İ����������Ȼ���⣬����Ҫ�����죩ʯ�ҡ���ʯ�ң���ʹ��̼��淋�ԭ����̼��識������ɰ���ͬʱ�����ɶ�����̼���壬��Ӧ�Ļ�ѧ����ʽΪ����NH4��2CO3$\frac{\underline{\;\;��\;\;}}{\;}$2NH3��+CO2��+2H2O��
�ʴ�Ϊ�������죩ʯ�ҡ���ʯ�ң���NH4��2CO3��2NH3��+CO2��+2H2O��
��4��װ��abc�ж���һ���ݻ��ϴ�������������ܹ����ս϶��Һ�壬�Ӷ�����ʹ������Һ�����ǰ���װ�ã����Է�ֹ������d�е����ܲ��뵽ˮ�У�������������ˮ���������������ܷ�������Ӧ�����Ȼ�̼���²㣬ˮ���ϲ㣬��ֹ�����������հ������ʴ�Ϊ��d��
��5������ͼ3��֪��Ӧ��Ϊ������һ�������Ͱ�������������Ϊ������ˮ������NO����ת��ΪN2��H2O��
�ʴ�Ϊ��N2��
��������һ�������Ͱ�����Ӧ���ɵ�����ˮ����Ӧ�а���ʧȥ�ĵ��ӵ����ʵ�������NO�������õ��ĵ��������ʵ�����2mol NH3ת��ΪN2ʧȥ6mol���ӣ�0.25mol O2�õ�1mol���ӣ���NOת��ΪN2�õ��ĵ���Ϊ2mol������NO�����ʵ���Ϊ1mol�������Ϊ22.4L��
�ʴ�Ϊ��22.4L��
���� ���⿼���˵����仯�������ʵķ�������ѧ����ʽ��д��ʵ��װ�õ����⡢������ԭ��Ӧ�����غ�ļ����֪ʶ�㣬��Ŀ�Ѷ��еȣ�

 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�| X | Y | ||
| Z | W | Q |
| A�� | ��Y��W�γɵ�����������ˮһ����ǿ�� | |
| B�� | X��Y���γ����ֳ����Ļ����� | |
| C�� | Z��Q�γɵ���������H2��һ�������·�Ӧ | |
| D�� | X��Y������ͬ�������� |
| A�� | ԭ��������a��b��c��d | B�� | ���Ӱ뾶��C��n+1��-��Dn-��A��n+1��+��Bn+ | ||
| C�� | ���ʻ�ԭ�ԣ�B��A�������ԣ�D��C | D�� | ԭ�Ӱ뾶��A��B��C��D |
���������գ�
��1��Rԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p2��X��������Ԫ������������Ӧ��ˮ�����У�������ǿ����HNO3��Z���⻯���ˮ��Һ�����ڵ�̲�����Q��R��U�ĵ����γɵľ��壬�۵��ɸߵ��͵�����˳����Si��Mg��Cl2��
��2��CuSO4������T4�ж��Ľⶾ������Ӧ������T����ۺ������ͭ���÷�Ӧ�Ļ�ѧ����ʽ��10CuSO4+P4+16H2O��4H3PO4+10Cu+10H2SO4��
��pHΪ4��5�Ļ����У�Cu2+��Fe2+�����ɳ�������Fe3+������ȫ��������ҵ����CuCl2�ǽ�Ũ�������������ȵ�80�����ң����������������ͭ����FeO������ֽ���ʹ֮�ܽ⣮
��3����ȥ����Һ���������ӵķ���������Һ��ͨ��Cl2���ټ���CuO��ĩ����pHΪ4��5��
̼����йػ�ѧ������������ʾ����Ҫ�����ͽ��������й���ʵ��
| ��ѧ�� | C-C | C-H | Si-Si | Si-H |
| ���ܣ�KJ/mol�� | 356 | 413 | 226 | 318 |
�������ڲ���Ԫ�ط�������۵����£�
| ������ | NaF | MgF2 | SiF4 |
| �۵�/K | 1266 | 1534 | 183 |
 CO��H2�Ļ�������׳ƺϳ�������һ����Ҫ�Ĺ�ҵԭ��������ҵ��������Ȼ������Ҫ�ɷ�ΪCH4����ˮ���и��������Ʊ��ϳ�����
CO��H2�Ļ�������׳ƺϳ�������һ����Ҫ�Ĺ�ҵԭ��������ҵ��������Ȼ������Ҫ�ɷ�ΪCH4����ˮ���и��������Ʊ��ϳ�������1���ü�����ˮ�����Ʊ��ϳ�����ÿ����1mol�ϳ�������Ҫ����51.5kJ�������÷�Ӧ���Ȼ�ѧ����ʽΪCH4��g��+H2O��g���TCO��g��+3H2��g����H=+206KJ/mol��
��2����һ���¶��£������Ϊ2L���ܱ������г���0.40mol CH4��0.60mol H2O��g����������Ӧ��CH4��g��+H2O��g��?CO��g��+3H2��g�������CH4��H2�����ʵ���Ũ����ʱ��仯���±���ʾ��
| Ũ�ȣ�mol/L�� ���� ʱ��/min | 0 | 1 | 2 | 3 | 4 |
| CH4 | 0.2 | 0.13 | 0.1 | 0.1 | 0.09 |
| H2 | 0 | 0.21 | 0.3 | 0.3 | 0.33 |
��3minʱ�ı�ķ�Ӧ�����������¶Ȼ�����H2O��g����Ũ�Ȼ��СCO��Ũ�ȣ�ֻ��һ�������ĸı伴�ɣ���
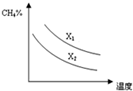
��3����֪һ��ѹǿ�£��¶ȡ�Ͷ�ϱ�X[$\frac{n��C{H}_{4}��}{n��{H}_{2}O��}$]�Ը÷�Ӧ��Ӱ����ͼ��ʾ��ͼ�е�����������ʾͶ�ϱȵĹ�ϵX1��X2���=������������������
| A�� | �����ԣ�X��Y | |
| B�� | ԭ�Ӱ뾶��Y��Z | |
| C�� | ����⻯������ȶ��ԣ�Z��W | |
| D�� | YԪ�������ﲻ����XԪ������������Ӧˮ�����ˮ��Һ |
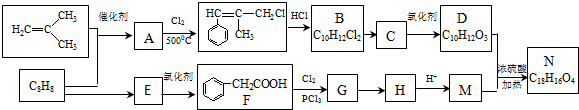
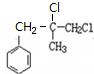 ��
��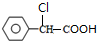 +2NaOH $��_{��}^{ˮ}$
+2NaOH $��_{��}^{ˮ}$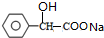 +NaCl+H2O���䷴Ӧ����Ϊˮ�ⷴӦ���кͷ�Ӧ��
+NaCl+H2O���䷴Ӧ����Ϊˮ�ⷴӦ���кͷ�Ӧ��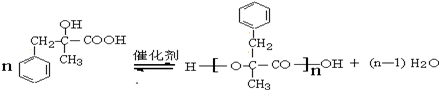 ��
��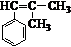 ��������
��������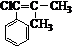 ��
��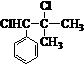 ��ԭ������Ϊ����������˫�������ļ��ϵ���ԭ�Ӹ���ȡ����
��ԭ������Ϊ����������˫�������ļ��ϵ���ԭ�Ӹ���ȡ����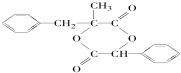 ��
��