��Ŀ����
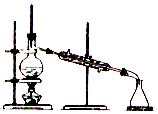
1����˾ƥ�֣�����ˮ���ᣬ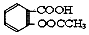 ����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128-135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣���Ӧԭ����ͼ��
����������Ӧ����㷺�Ľ��ȡ���ʹ�Ϳ���ҩ������ˮ���������ֽ⣬�ֽ��¶�Ϊ128-135�森ijѧϰС����ʵ������ˮ���ᣨ���ǻ������ᣩ�������[��CH3CO��2O]Ϊ��Ҫԭ�Ϻϳɰ�˾ƥ�֣���Ӧԭ����ͼ��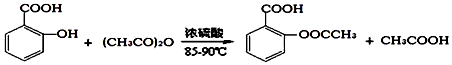
����������ͼ��
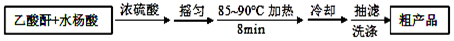
��Ҫ�Լ��Ͳ�Ʒ������������
| �� �� | ��Է������� | �۵��е㣨�棩 | ˮ |
| ˮ���� | 138 | 158���۵㣩 | �� |
| ������ | 102 | 139.4���е㣩 | ��ˮ�� |
| ����ˮ���� | 180 | 135���۵㣩 | �� |
��1���Ʊ���˾ƥ��ʱ��Ҫʹ�ø����������ԭ���Ǵ�������ˮ�⣮
��2���ϳɹ���������ʵļ��ȷ�����ˮԡ���ȣ�
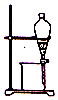
��3���ᴿ�ֲ�Ʒ�������£����Ȼ�����װ����ͼ��ʾ��
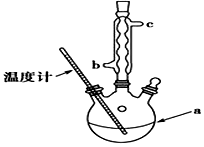
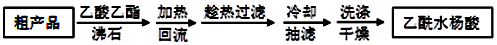
��a����������������ƿ������ˮ������������c���b����c������
�ڳ��ȹ��˵�ԭ���Ƿ�ֹ����ˮ����ᾧ������
�ۼ������ղ�Ʒ���Ƿ���ˮ����Ļ�ѧ������ȡ�����ᾧ���Թ��У�������ˮ�ܽ⣬�μ�FeCl3��Һ����������ɫ��ˮ���ᣮ
������˵����ȷ����ab��
a�������ᴿ�������������������������ܼ�
b�������ᴿ�ֲ�Ʒ�ķ������ؽᾧ
c�����������ᴿ���̿��Եó���˾ƥ�������������е��ܽ�ȵ���ʱ��
��4����ʵ����ԭ��������2.0gˮ���ᡢ5.0mL����������=1.08g/cm3�������ճ�����Ʒ����Ϊ2.2g������������ˮ����IJ���Ϊ84.3%�����ðٷ�����ʾ��������С�����һλ��
���� ��1��������������ˮ�⣬Ӧ�ø���������
��2�������¶���85�桫90�棬�¶ȵ���100�棬Ӧ��ȡˮԡ���ȣ�
��3����aΪ������ƿ������ʱ��Ӧ����ʹ��ˮ���������ܣ�
������ˮ�������¶Ƚϵ�ʱ�ܽ�Ƚ�С��Ӧ���ȹ��ˣ�
�ۿ��ü�����ǻ��ķ������飻
�������̿�֪���������������������ܼ������ã������ȼ��Ⱥ������ķ������룬Ϊ�ؽᾧ�ķ�����
��4������������������ˮ������������ɼ�������ʣ�
��� �⣺��1��������������ˮ���������ᣬ������Ӧ�����ֹ������ˮ�⣬
�ʴ�Ϊ����������ˮ�⣻
��2�������¶���85�桫90�棬С��100�棬Ӧʹ��ˮԡ���ȣ�
�ʴ�Ϊ��ˮԡ���ȣ�
��3����aΪ������ƿ������ʱ��Ӧ����ʹ��ˮ���������ܣ�������ˮ������������c����b�ڽ��룬�ʴ�Ϊ��������ƿ�� c��
������ˮ�������¶Ƚϵ�ʱ�ܽ�Ƚ�С��Ӧ���ȹ��ˣ��ɷ�ֹ����ˮ����ᾧ�������ʴ�Ϊ����ֹ����ˮ����ᾧ������
��ˮ���Ậ�з��ǻ��������Ȼ���������ɫ��Ӧ�����鷽����ȡ�����ᾧ���Թ��У�������ˮ�ܽ⣬�μ�FeCl3��Һ����������ɫ��ˮ���ᣬ
�ʴ�Ϊ��ȡ�����ᾧ���Թ��У�������ˮ�ܽ⣬�μ�FeCl3��Һ����������ɫ��ˮ���
��a���������������������ܼ������ã���������ˮ������ܽ⣬����ȷ��
b�������ȼ��Ⱥ������ķ������룬Ϊ�ؽᾧ�ķ���������ȷ��
c������˾ƥ�������������е��ܽ�ȵ���ʱ��С���ʴ���
�ʴ�Ϊ��ab��
��4��n��ˮ���ᣩ=$\frac{20g}{138g/mol}$=0.0145mol��n����������=$\frac{5mL��1.08g/mL}{102g/mol}$=0.053mol������������������ˮ����0.0145mol������Ϊ$\frac{2.2g}{0.0145mol��180g/mol}$=84.3%��
�ʴ�Ϊ��84.3%��
���� ���⿼���л���ϳɵķ�����������ۣ�Ϊ�߿��������ͣ����ؿ���ѧ���ķ���������ʵ�������ͼ�����������Ŀ�Ѷ��еȣ�����ע�����ʵ�����ԭ�������������Ϣ������ʵ�����������ѧϰ��ע����ۣ�

 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�| A�� | ��ɫ��dz | B�� | ���ɫ | C�� | û�иı� | D�� | ���ػ�ɫ |
| A�� | NH2- | B�� | Cl | C�� | NH3 | D�� | Na+ |
| ���� | ���� | ���� | |
| A | ������̪��Na2CO3��Һ��BaCl2��Һ��� | ��ɫ��dz | Na2CO3��Һ�д���ˮ��ƽ�� |
| B | ��Cl2ͨ��I2������Һ�� | ��Һ��ɫ��ȥ | Cl2��Ư���� |
| C | ��Ũ�ȵ�NaCl��KI��Һ�еμ�����AgNO3��Һ | ���ֻ�ɫ���� | Ksp��AgCl����Ksp��AgI�� |
| D | �Թ��м����������ۺ�ϡ���ᣬ��ȴ�����NaOH��Һ�к������ԣ��ټ����ˮ | ��Һδ���� | ���ۼ���ȫˮ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
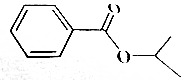 ij�л���ṹ��ͼ��ʾ���йظ����ʵ�������ȷ���ǣ�������
ij�л���ṹ��ͼ��ʾ���йظ����ʵ�������ȷ���ǣ�������| A�� | ������������ˮ���������Ҵ������� | |
| B�� | �������ܷ����ӳɡ�������ˮ��ȷ�Ӧ | |
| C�� | 1mol�������������4mol�����ӳ� | |
| D�� | ����������̼ԭ�ӿ��ܹ�ƽ�� |
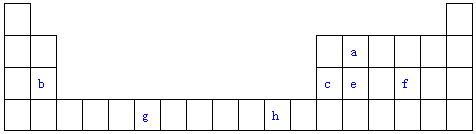
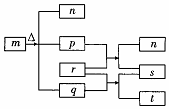 ������Ԫ��W��X��Y��Z��ԭ�������������ӣ�m��p��r������ЩԪ����ɵĶ�Ԫ��������³�ѹ�£�pΪ��ɫ��ζ���壬qΪҺ̬��rΪ����ɫ���壬s�����嵥�ʣ��������ʵ�ת����ϵ��ͼ��ʾ������˵��������ǣ�������
������Ԫ��W��X��Y��Z��ԭ�������������ӣ�m��p��r������ЩԪ����ɵĶ�Ԫ��������³�ѹ�£�pΪ��ɫ��ζ���壬qΪҺ̬��rΪ����ɫ���壬s�����嵥�ʣ��������ʵ�ת����ϵ��ͼ��ʾ������˵��������ǣ�������| A�� | ԭ�Ӱ뾶��Z��Y��X | B�� | �����̬�⻯����ȶ��ԣ�Y��X | ||
| C�� | m��n��r���������Ӽ����ۼ� | D�� | m��n��t�����ƻ�ˮ�ĵ���ƽ�� |
��1�������Ƽ�ѭ�������ѳ������е�SO2��
�����Ƽ�ѭ�����У�Na2SO3��Һ����Ϊ����Һ������NaOH��Һ����SO2�Ƶã��÷�Ӧ�����ӷ���ʽ��2OH-+SO2=SO32-+H2O��
������Һ����SO2�Ĺ����У�pH��m��SO32-����m��HSO3-���仯��ϵ���±���
| n��SO32-����n��HSO3-�� | 91��9 | 1��1 | 9��91 |
| pH | 8.2 | 7.2 | 6.2 |
�۵�����Һ��pH����ԼΪ6ʱ����������������������ʾ��ͼ���£�

д��HSO3-�������ŵ�ĵ缫��Ӧʽ��HSO3-+H2O-2e-=SO42-+3H+��������������ҺpH����8����ʱ������Һ������ѭ�����ã�
��2����CH4����ԭNO2������������������Ⱦ�����磺
CH4��g��+4NO2��g���T4NO��g��+CO2��g��+2H2O����H=-574kJ•mol-1
CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O����H=-1160kJ•mol-1
���ñ�״����4.48LCH4��ԭNO2��N2������������ת�Ƶĵ�������Ϊ1.60NA����1.6NA��������٤��������ֵ��NA��ʾ�����ų�������Ϊ173.4kJ��
��3����ҵ�Ϻϳɰ������������Ʊ������У����е�һ����ӦΪ��
CO��g��+H2O��g��$\frac{\underline{\;����\;}}{\;}$CO2��g��+H2��g������H��0
һ�������£���CO��g����H2O��g���������Ϊ1��2�����ܱ������з���������Ӧ���ﵽƽ��ʱ���CO��g����H2O��g�������Ϊ1��6����ƽ�ⳣ��K=2.67��������������λС������