��Ŀ����
�Ͻ����������������ɽ����������;�dz��㷺��
�ƼغϽ���ں˷�Ӧ�����������ȼ���5.05g�ƼغϽ�����200mLˮ����0.075mol������
��1 ��������Һ�����������ӵ����ʵ���Ũ�ȣ�������Һ����仯����
��2 �����㲢ȷ������-�غϽ�Ļ�ѧʽ ��
þ���Ͻ�㷺���ڻ���������ͷɻ�����ҵ�ȣ�ȡ��ͬ������þ���Ͻ���Ʒ�ֱ��30ml��ͬŨ�ȵ����ᷴӦ����ȡ�Ͻ�����������������������£����±���ʾ��
��3 ������þ���Ͻ���þ��������֮�ȣ�
�ƼغϽ���ں˷�Ӧ�����������ȼ���5.05g�ƼغϽ�����200mLˮ����0.075mol������
��1 ��������Һ�����������ӵ����ʵ���Ũ�ȣ�������Һ����仯����
��2 �����㲢ȷ������-�غϽ�Ļ�ѧʽ
þ���Ͻ�㷺���ڻ���������ͷɻ�����ҵ�ȣ�ȡ��ͬ������þ���Ͻ���Ʒ�ֱ��30ml��ͬŨ�ȵ����ᷴӦ����ȡ�Ͻ�����������������������£����±���ʾ��
| ʵ����� | A | B | C |
| �Ͻ�����/mg | 510 | 765 | 918 |
| �������/mL | 560 | 672 | 672 |
���㣺�йػ���ﷴӦ�ļ���
ר�⣺������
��������1��������Ӧ��2Na+2H2O=2NaOH+H2����2K+2H2O=2KOH+H2�����ɷ���ʽ��֪n��OH-��=2n��H2�����ٸ���c=
������Һ������������Ũ�ȣ�
��2���������Ͻ���NaΪamol��KΪbmol�����ݶ����������������������ʵ����з��̼��㣬����ȷ���Ͻ�Ļ�ѧʽ��
��3�����ж�A�н�����ȫ��Ӧ��Ȼ����ݻ�Ͻ������������������������ʵ�����ʽ���㣮
| n |
| V |
��2���������Ͻ���NaΪamol��KΪbmol�����ݶ����������������������ʵ����з��̼��㣬����ȷ���Ͻ�Ļ�ѧʽ��
��3�����ж�A�н�����ȫ��Ӧ��Ȼ����ݻ�Ͻ������������������������ʵ�����ʽ���㣮
���
�⣺��1��������Ӧ��2Na+2H2O=2NaOH+H2����2K+2H2O=2KOH+H2�����ɷ���ʽ��֪n��OH-��=2n��H2��=0.075mol��2=0.15mol��
����Һ��c��OH-��=
=0.75mol/L��
����Һ�����������ӵ�Ũ��Ϊ0.75mol/L��
��2���������Ͻ���NaΪxmol��KΪymol����x+y=0.075��2��23x+39y=5.05��
��ã�x=0.05��y=0.1��
��Na��K�����ʵ���֮��=0.05mol��0.1mol=1��2���ʸúϽ�Ļ�ѧʽΪNaK2��
�ʴ�Ϊ��NaK2��
��3�����ݱ������ݿ�֪��A�н�����ȫ��Ӧ�����������
��Ͻ���Mg��Al�����ʵ����ֱ�Ϊxmol��ymol�����У�
24x+27y=0.510g
x+
y=
=0.025mol����ã�x=y=0.01 mol��
�Ͻ���þ��������֮��Ϊ��24g/mol��0.01mol��27g/mol��0.01mol=8��9��
�ʴ�Ϊ��8��9��
����Һ��c��OH-��=
| 0.15mol |
| 0.2L |
����Һ�����������ӵ�Ũ��Ϊ0.75mol/L��
��2���������Ͻ���NaΪxmol��KΪymol����x+y=0.075��2��23x+39y=5.05��
��ã�x=0.05��y=0.1��
��Na��K�����ʵ���֮��=0.05mol��0.1mol=1��2���ʸúϽ�Ļ�ѧʽΪNaK2��
�ʴ�Ϊ��NaK2��
��3�����ݱ������ݿ�֪��A�н�����ȫ��Ӧ�����������
��Ͻ���Mg��Al�����ʵ����ֱ�Ϊxmol��ymol�����У�
24x+27y=0.510g
x+
| 3 |
| 2 |
| 0.560L |
| 22.4L/mol |
�Ͻ���þ��������֮��Ϊ��24g/mol��0.01mol��27g/mol��0.01mol=8��9��
�ʴ�Ϊ��8��9��
���������⿼���˻���ﷴӦ�ļ��㣬��Ŀ�Ѷ��еȣ�����֪ʶ��϶ࡢ�������ϴ�ֿ���ѧ���Ļ�ѧ����������ע������غ㡢�����غ㶨���ڻ�ѧ�����е�Ӧ�÷�����

��ϰ��ϵ�д�
 �������ϵ�д�
�������ϵ�д�
�����Ŀ
��ͼ��NaCl��MgSO4���ܽ�����ߣ�����˵����ȷ���ǣ�������
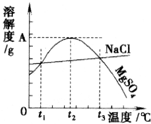

| A��ֻ����t1��ʱ��NaCl��MgSO4���ܽ�Ȳ���� |
| B��t1�桫t2�棬MgSO4���ܽ�����¶����߶����� |
| C����t2��ʱ��MgSO4��Һ����������������� |
| D����MgSO4������Һ���¶ȴ�t3�潵��t2��ʱ���о������� |
�백���Ƚϣ������Ƽ��ͻ�����ŵ��ǣ�������
| A���豸�� |
| B��ԭ���� |
| C��ѭ�����õ����ʶ� |
| D��NaCl�����ʸ� |
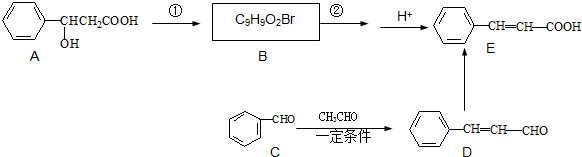
 ���л���A��ͬ���칹�壬����˵���������
���л���A��ͬ���칹�壬����˵���������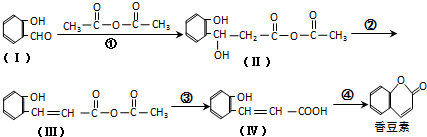
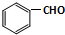 ��CH3CHO����֮���ܷ������Ƣ١��ڵ�������Ӧ���������л���Ľṹ��ʽΪ
��CH3CHO����֮���ܷ������Ƣ١��ڵ�������Ӧ���������л���Ľṹ��ʽΪ
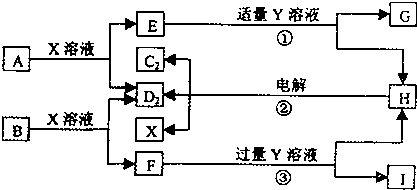
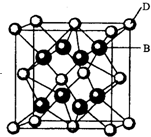 ��A��B��C��D��E��F����Ԫ�أ�A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�ǵ縺������Ԫ�أ�C��2p�����������δ�ɶԵĵ����ӣ�Fԭ�Ӻ����������B��C���������֮�ͣ�D������Ԫ������Eͬ���ڣ�E���γɺ�ɫ����ש��ɫ����E2O�ͺ�ɫ��EO���������D��B���γ����ӻ������侧���ṹ����ͼ��ʾ����ش��������⣮
��A��B��C��D��E��F����Ԫ�أ�A�����ڱ���ԭ�Ӱ뾶��С��Ԫ�أ�B�ǵ縺������Ԫ�أ�C��2p�����������δ�ɶԵĵ����ӣ�Fԭ�Ӻ����������B��C���������֮�ͣ�D������Ԫ������Eͬ���ڣ�E���γɺ�ɫ����ש��ɫ����E2O�ͺ�ɫ��EO���������D��B���γ����ӻ������侧���ṹ����ͼ��ʾ����ش��������⣮