��Ŀ����
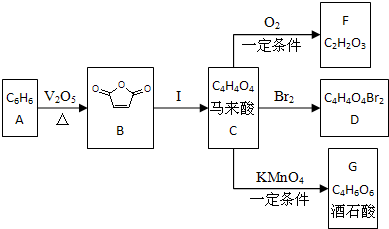
�����ᣨC4H4O4���Ǻϳɲ����;�����֬����Ҫԭ�ϣ�������ú̿�������C6H6Ϊԭ���Ƶã�
��1��д�����������ƣ�A ��F ��
��2��B���Ҷ����ڴ��������������������C10H10O8��д����Ӧ����ʽ�� ��
��3��д��B����Ҷ���������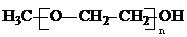 ��Ӧ���ɲ���Ľṹ��ʽ����֪�Ѽ���ѧ�����ȶ����� ��
��Ӧ���ɲ���Ľṹ��ʽ����֪�Ѽ���ѧ�����ȶ����� ��
��4��д����������Ҫ������ʵĽṹ��ʽ��һ�֣��������ͬ���칹���У���������2���Ȼ� ����������1���Ȼ� ��
��5��д��Dת��ΪG�Ļ�ѧ����ʽ ��

��1��д�����������ƣ�A
��2��B���Ҷ����ڴ��������������������C10H10O8��д����Ӧ����ʽ��
��3��д��B����Ҷ���������
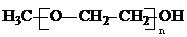 ��Ӧ���ɲ���Ľṹ��ʽ����֪�Ѽ���ѧ�����ȶ�����
��Ӧ���ɲ���Ľṹ��ʽ����֪�Ѽ���ѧ�����ȶ�������4��д����������Ҫ������ʵĽṹ��ʽ��һ�֣��������ͬ���칹���У���������2���Ȼ�
��5��д��Dת��ΪG�Ļ�ѧ����ʽ
������C6H6��ú�ĸ�������AΪ��������������Ӧ����B��B��Ӧ����C������B�Ľṹ��ʽ��C�ķ���ʽ֪��C�Ľṹ��ʽΪHOOCCH=CHCOOH��C����������F��F�IJ����Ͷ�=
=2�����F�ķ���ʽ֪��F�Ľṹ��ʽΪOHCCOOH��C���巢���ӳɷ�Ӧ����D��D�Ľṹ��ʽΪHOOCCHBrCHBrCOOH��C�����������������G�����G�ķ���ʽ֪��G�Ľṹ��ʽΪ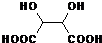 ��
��
| 2��2+2-2 |
| 2 |
 ��
������⣺C6H6��ú�ĸ�������AΪ��������������Ӧ����B��B��Ӧ����C������B�Ľṹ��ʽ��C�ķ���ʽ֪��C�Ľṹ��ʽΪHOOCCH=CHCOOH��C����������F��F�IJ����Ͷ�=
=2�����F�ķ���ʽ֪��F�Ľṹ��ʽΪOHCCOOH��C���巢���ӳɷ�Ӧ����D��D�Ľṹ��ʽΪHOOCCHBrCHBrCOOH��C�����������������G�����G�ķ���ʽ֪��G�Ľṹ��ʽΪ ��
��
��1��ͨ�����Ϸ���֪��A�DZ���F�Ľṹ��ʽΪOHCCOOH�����Ա��к��б�����F�й�����������ȩ�����Ȼ���
�ʴ�Ϊ��������ȩ�����Ȼ���
��2��B���Ҷ����ڴ��������·�Ӧ�����������C10H10O8��
��Ӧ����ʽΪ��2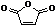 +HO-CH2-CH2-OH
+HO-CH2-CH2-OH
HOOCCH=CHCOOCH2CH2OOCCH=CHCOOH��
�ʴ�Ϊ��2 +HO-CH2-CH2-OH
+HO-CH2-CH2-OH
HOOCCH=CHCOOCH2CH2OOCCH=CHCOOH��
��3��B����Ҷ���������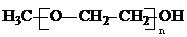 ��Ӧ���ɲ���Ľṹ��ʽΪ HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3����
��Ӧ���ɲ���Ľṹ��ʽΪ HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3����
�ʴ�Ϊ��HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3����
��4��C�Ľṹ��ʽΪHOOCCH=CHCOOH���������ͬ���칹���У���������2���Ȼ��Ľṹ��ʽΪ��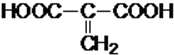 ����������1���Ȼ��Ľṹ��ʽΪ
����������1���Ȼ��Ľṹ��ʽΪ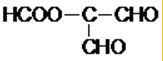 ��
��
�ʴ�Ϊ��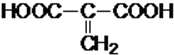 ��
��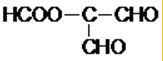 ��
��
��5��D�Ľṹ��ʽΪHOOCCHBrCHBrCOOH��G�Ľṹ��ʽΪ ��D���������Ƶ�ˮ��Һ����ȡ����Ӧ����G����Ӧ����ʽΪ
��D���������Ƶ�ˮ��Һ����ȡ����Ӧ����G����Ӧ����ʽΪ
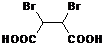 +2H2O
+2H2O
 +2HBr��
+2HBr��
�ʴ�Ϊ�� +2H2O
+2H2O
 +2HBr��
+2HBr��
| 2��2+2-2 |
| 2 |
 ��
����1��ͨ�����Ϸ���֪��A�DZ���F�Ľṹ��ʽΪOHCCOOH�����Ա��к��б�����F�й�����������ȩ�����Ȼ���
�ʴ�Ϊ��������ȩ�����Ȼ���
��2��B���Ҷ����ڴ��������·�Ӧ�����������C10H10O8��
��Ӧ����ʽΪ��2
 +HO-CH2-CH2-OH
+HO-CH2-CH2-OH | ���� |
�ʴ�Ϊ��2
 +HO-CH2-CH2-OH
+HO-CH2-CH2-OH | ���� |
��3��B����Ҷ���������
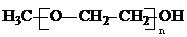 ��Ӧ���ɲ���Ľṹ��ʽΪ HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3����
��Ӧ���ɲ���Ľṹ��ʽΪ HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3�����ʴ�Ϊ��HOOCCH=CHCOO��CH2CH2O��nCH3����H3C��OCH2CH2��nOOCCH=CHCOO��CH2CH2O��nCH3����
��4��C�Ľṹ��ʽΪHOOCCH=CHCOOH���������ͬ���칹���У���������2���Ȼ��Ľṹ��ʽΪ��
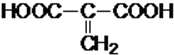 ����������1���Ȼ��Ľṹ��ʽΪ
����������1���Ȼ��Ľṹ��ʽΪ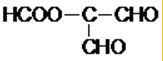 ��
���ʴ�Ϊ��
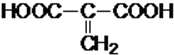 ��
��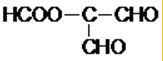 ��
����5��D�Ľṹ��ʽΪHOOCCHBrCHBrCOOH��G�Ľṹ��ʽΪ
 ��D���������Ƶ�ˮ��Һ����ȡ����Ӧ����G����Ӧ����ʽΪ
��D���������Ƶ�ˮ��Һ����ȡ����Ӧ����G����Ӧ����ʽΪ +2H2O
+2H2O | NaOH |
| �� |
 +2HBr��
+2HBr���ʴ�Ϊ��
 +2H2O
+2H2O | NaOH |
| �� |
 +2HBr��
+2HBr�����������⿼�����л�����ƶϣ���ȷ�л���Ĺ����ż��������ǽⱾ��ؼ��������������ϵķ��������ƶϣ��ѵ���B�ֱ���Ҷ��������Ҷ��������ѷ�Ӧ����ʽ����д���Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ