��Ŀ����
20�����ܸ���ĸ��ֲ��ϵĹ㷺Ӧ�ô����������ǵ�����ˮƽ���ش����⣺�ټ�ͥ�С��־������������Ͻ����ɵģ����ղ�ʱ�ܷ�Ŵ������������ܻ��
�����й�������Ʒ������ʩ��˵���в���ȷ����B������ţ�
A�������г���Ȧ�϶���һ���������Ħ����λ���ϻ��ͻ����
B���Ʋ�����ʱ����ͬ�����µ������������������������������п������������Ϊ����
C���ں��ֵ�����ϳ�����п�飬�Ҷ��ڸ���
D����������Ƴɵ��ߺͲ;ߣ��еĵ������п�������
�۸�������������о۰���������ĭ���۰�������C������ĸ����
A���������� B�����ǽ������� C���л��߷��Ӳ���
��ú�м���ʯ��ʯ���Դ�����SO2���ŷţ�������Ӧ�Ļ�ѧ����ʽ��2CaCO3+2SO2+O2$\frac{\underline{\;����\;}}{\;}$2CaSO4+2CO2��
���� ���������������ﶼ�ܺʹ��ᷴӦ�����Σ�
��A������������ˮ�ܷ�ֹ������ʴ��
B����ԭ����У��������Ľ������ٱ���ʴ���������Ľ�����������
C����ԭ����У��������Ľ������ٱ���ʴ���������Ľ�����������
D���ı����ʽṹ�ܷ�ֹ��������ʴ��
���л��߷��ӻ������Ƹ߷��ӻ������߷��ӣ��ֳƸ߾���߷��ӻ�������Է��������ܴ�һ����10000���ϣ�
����ú�м���ʯ��ʯ��Ϊ����������Լ���SO2���ŷţ�����CaSO4
��� �⣺���������������ﶼ�ܺʹ��ᷴӦ�����Σ��������Ͻ����ղ�ʱ���ܷŴ���������������ȡ����������������մ�֢���ʴ�Ϊ����
��A�������г���Ȧ�϶���һ���������Ħ����λ���ϻ��ͻ���ͣ��ܸ���������ˮ�������ܷ�ֹ����ʴ����A��ѡ��
B����ͬ�����£�������������������γ�ԭ���ʱFe���������ٱ���ʴ�������������п���γ�ԭ���ʱFe����������������������Ϊ���ã���Bѡ��
C���ں��ֵ�����ϳ�����п��ʱ��Fe��Zn�ͺ�ˮ����ԭ��أ�������������������C��ѡ��
D����������Ƴɵ��ߺͲ;ߣ��еĵ������п����������ı�����ṹ������������ˮ�������ܷ�ֹ������ʴ����D��ѡ��
��ѡB��
�۾۰������ڸ߷��Ӻϳɲ��ϣ����л��߷��ӻ�����ʴ�Ϊ��C��
��ú̿�е���ȼ�ղ�����SO2���γ��������������Ҫ���أ���ú�м���ʯ��ʯ��Ϊ����������Լ���SO2���ŷţ�����CaSO4��
��Ӧ�ķ���ʽΪ��2CaCO3+2SO2+O2$\frac{\underline{\;����\;}}{\;}$2CaSO4+2CO2���ʴ�Ϊ��2CaCO3+2SO2+O2$\frac{\underline{\;����\;}}{\;}$2CaSO4+2CO2��
���� ���⿼�������ʴ���������ȷ������ʴԭ���ǽⱾ��ؼ���֪����ֹ������ʴ�ķ����У�ͿĨ���ᡢ�ı�����ṹ����ԭ��������������������Ŀ�ѶȲ���

��1�������ʵķ������д����Ŀհ״��������ţ�
| ����� | �ܵ�������� | ����� | �ǵ���� |
| ���ڸ�������� |
��3������ߺ͢�ķ����Ƕ����ЧӦ������μӵ�������������ʵ���������Ȳ������ɫ������������ܽ�
��4�������ͨ�����ǡ����ȫ��Ӧ�����ĵ��������ı仯Ϊ������ǿ
��5��������Һ�μӵ��ݵ���Һ����ǡ�ó���Ϊֹʱ�����ӷ���ʽΪBa2++OH-+H++SO42-�TBaSO4��+H2O��
| A�� | c��HCO3-����c��H2CO3�� | B�� | c��OH-��=2c��H+�� | ||
| C�� | c��Na+��=2c��CO32-�� | D�� | c��Na+����c��CO32-��+c��HCO3-�� |
| A�� | ��Ba��OH��2��Һ����μ���NH4HSO4��Һ���պó�����ȫ��Ba2++2OH-+H++SO42-+NH4+=BaSO4��+NH3•H2O+H2O | |
| B�� | ���Ƶ�ˮ�⣺S2-+2H2O?H2S+2OH- | |
| C�� | ��Fe��NO3��3��Һ�м��������HI��Һ��Fe3++3NO3-+10I-+12H+=5I2+Fe2++3NO��+6H2O | |
| D�� | NaHCO3��Һ�еμ���������ʯ��ˮ��2HCO3-+Ca2++2OH-=CaCO3��+2H2O+CO32- |
| A�� |  | B�� |  | C�� |  | D�� |  |
| A�� | ǿ����ʵ�ˮ��Һһ�������������Һ�ĵ�������ǿ | |
| B�� | ǿ����ʶ������ӻ����������ʶ��ǹ��ۻ����� | |
| C�� | ǿ����ʵ�ˮ��Һ�в��������ʷ��� | |
| D�� | �������ζ���������ʣ��������ζ���ǿ����� |
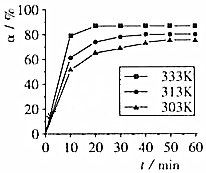 ���仯�����ڹ�ҵ������ҪӦ�ã���֪���¼�����Ӧ��
���仯�����ڹ�ҵ������ҪӦ�ã���֪���¼�����Ӧ����BCl3��g��+3H2O��I��=H3BO3��s��+3HCl��g����H1
��B2O3��s��+3C��s��+3Cl2��g��?2BCl3��g��+3CO��g����H2
��H3BO3��aq��+H2O��I��?[B��OH��4]-��aq��+H+��aq��
����H3BO3��I��+3CH3OH��I��?B��OCH3��3��I��+3H2O��I��
��1����֪����Ľṹ�ǣ�
 ����Ӧ������ػ�ѧ�������������£�
����Ӧ������ػ�ѧ�������������£�| ��ѧ�� | H-O | H-Cl | B-O | B-Cl |
| E��KJ•mol-1�� | 465 | 431 | 809 | 427 |
��2����֪��H2��0������ʹ��Ӧ��ƽ��������Ӧ�����ƶ������ܼӿ췴Ӧ���ʵĴ�ʩ��bc��
a����ѹ b������ c������Cl2��Ũ�� d�����߲���BCl3 e�������ʵ��Ĵ��� f������̿����
��3��д����Ӧ���ƽ�ⳣ������ʽK=$\frac{[[B��OH��{\;}_{4}]{\;}^{-}][H{\;}^{+}]}{[{H}_{3}B{O}_{3}]}$��д��������[B��OH��4]-�ĵ���ʽ��
 ��
����4���ڷ�Ӧ���У�H3BO3��ת�����ڲ�ͬ�¶����淴Ӧʱ�䣨t���ı仯��ͼ����T=333K����ʼc��H3BO3��=1mol•L-1����0��10min�ķ�Ӧ����v��H3BO3��=0.08mol/��L•min����40min�ﵽƽ��ı��¶�ΪT��c��H3BO3 ����0.005mol•L-1•min-1��ƽ�����ʽ��ͣ�����10min�ִﵽƽ�⣬��T��333K���á�����������������=����գ����¶ȶԸ÷�Ӧ�����ʺ�ƽ���ƶ���Ӱ���������¶ȷ�Ӧ���ʼӿ죬ƽ���������ƶ���