��Ŀ����
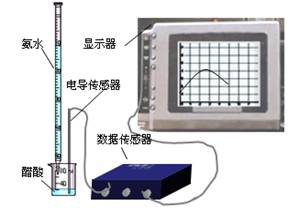
DISϵͳ�����ֻ���Ϣϵͳ�����ɴ����������ݲɼ����ͼ������ɣ�ijѧϰС����DISϵͳ�ⶨʳ�ð״��д�������ʵ���Ũ�ȣ�����Һ�ĵ����������жϵζ��յ㡣ʵ�鲽�����£�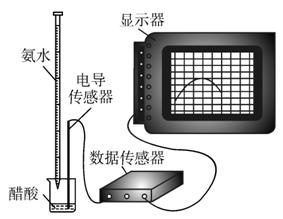
(1)��________(����������)��ȡ10.00 mL��ʳ�ð״ף���________(����������)����ˮϡ�ͺ�ת�Ƶ�100 mL____________(����������)�ж��ݣ�Ȼ��ϡ�ͺ����Һ�����Լ�ƿ�С�
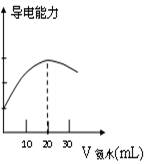
(2)��ȡ20.00 mL��������Һ�����ձ��У����Ӻ�DISϵͳ(����ͼ)�����ձ��еμ�Ũ��Ϊ0.100 0 mol/L�İ�ˮ���������Ļ����ʾ����Һ���������氱ˮ����仯������(��ͼ1-2-12)��

ͼ1-2-12
���õζ���ʢ��ˮǰ���ζ���Ҫ��_______��ϴ2��3�飬��ϴ��Ŀ����_________________��
�ڰ�ˮ����ᷴӦ�����ӷ���ʽ��___________________________________________��
��ʳ�ð״��д�������ʵ���Ũ����__________________________________________��
����:������Ҫ����Һ�����ȡ��ϡ�ͼ��к͵ζ��Ȼ���������������ĿҪ����ȡ����ľ�ȷ��Ϊ0.01 mL���������ζ��ܻ���Һ������ȡ�����ձ���ϡ�ͺ���ת��������ƿ�в���ˮ���̶ȣ���ɵõ�������Һ��Ϊȷ���ζ�������ʢҺ���Ũ����ԭ��Һһ�£�һ�����ô�װҺ����ϴ2��3�Ρ�����������ͼ��֪��V(NH3��H2O)=20 mLʱ����ǡ����ȫ��Ӧ����CH3COOH+NH3��H2O=CH3COO-+![]() +H2O�ɵã�
+H2O�ɵã�
c��(HAc)��20��10-3 L=20��10-3��0.100 0 mol��L-1,c��(HAc)=0.100 0 mol��L-1������ԭ�����Ũ��Ϊ1.000 mol��L-1��
��:(1)�ζ���(����Һ��) �ձ� ����ƿ
(2)��0.1 000 mol��L-1�İ�ˮ ȷ����Ӧʱ��ˮ��Ũ��Ϊ0.1 000 mol��L-1
��CH3COOH+NH3��H2O![]() CH3COO-+
CH3COO-+![]() +H2O
+H2O
��1.000 mol��L-1

 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�