��Ŀ����
16������������ݣ�����ѡ����ȷ���ǣ�������| ��ѧʽ | CH3COOH | H2SO3 | NH3•H2O |
| ���볣��K��25�棩 | Ka=1.8��10-5 | Ka1=1.23��10-2��Ka2=6.6��10-8 | Kb=1.8��10-5 |
| A�� | Na2SO3��Һ�еμ��������ᣬ��Ӧ�����ӷ���ʽΪ��SO32-+2CH3COOH=SO2��+H2O+2CH3COO- | |
| B�� | 25��ʱ��pH=3�Ĵ����pH=11�İ�ˮ�������Ϻ��Һ��ˮ�ĵ��뱻�ٽ� | |
| C�� | NaHSO3��Һ�����ԣ�����ΪKw/Ka1��Ka2 | |
| D�� | 0.1mol/L��CH3COOH��Һ���Ũ�ȵ������CH3COONa��Ϻ���Һ�е����������¹�ϵ��c��H+��+c��CH3COOH��=c��CH3COO-��+c��OH-�� |
���� A���������һ���������Դ��ڴ��
B.25��ʱ��pH=3�Ĵ����pH=11�İ�ˮ�������Ϻ��߷�Ӧ���ɴ��ᰱ��笠�����ˮ�������ԣ����������ˮ���Լ��ԣ�
C��NaHSO3��Һ�д���ˮ��ƽ�⣬HSO3-+H2O?H2SO3+OH-��K=$\frac{c��O{H}^{-}��c��{H}_{2}S{O}_{3}��}{c��HS{{O}_{3}}^{-}��}$=$\frac{c��O{H}^{-}��c��{H}_{2}S{O}_{3}��}{c��HS{{O}_{3}}^{-}��}$��$\frac{c��{H}^{+}��}{c��{H}^{+}��}$=$\frac{Kw}{K{a}_{1}}$��
D��CH3COOH��CH3COONa�Ļ����Һ��ֻ��4�����ӣ��ݵ���غ���c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�Ϊ��c��CH3COOH��+c��CH3COO-��=2c��Na+������������жϣ�
��� �⣺A���������һ���������Դ��ڴ��ᣬ�������ɶ����������壬Na2SO3��Һ�еμ��������ᣬ��Ӧ�����ӷ���ʽΪ��SO32-+CH3COOH=HSO3-+CH3COO-����A����
B.25��ʱ��pH=3�Ĵ����pH=11�İ�ˮ�������Ϻ��߷�Ӧ���ɴ��ᰱ��笠�����ˮ�������ԣ����������ˮ���Լ��ԣ�����ˮ��ٽ������Һ��ˮ�ĵ��뱻�ٽ�����B��ȷ��
C��NaHSO3��Һ�д���ˮ��ƽ�⣬HSO3-+H2O?H2SO3+OH-��K=$\frac{c��O{H}^{-}��c��{H}_{2}S{O}_{3}��}{c��HS{{O}_{3}}^{-}��}$=$\frac{c��O{H}^{-}��c��{H}_{2}S{O}_{3}��}{c��HS{{O}_{3}}^{-}��}$��$\frac{c��{H}^{+}��}{c��{H}^{+}��}$=$\frac{Kw}{K{a}_{1}}$��NaHSO3��Һ�����ԣ��������ˮ�⣬Kw/Ka1��Ka2����C����
D��CH3COOH��CH3COONa�Ļ����Һ��ֻ��4�����ӣ��ݵ���غ���c��Na+��+c��H+��=c��CH3COO-��+c��OH-���������غ�Ϊ��c��CH3COOH��+c��CH3COO-��=2c��Na+����2c��H+��+c��CH3COOH��=c��CH3COO-��+2c��OH-������D����
��ѡB��
���� ���⿼����������ʵ���ƽ���Ӱ�����ء���Ӧ������жϡ�ˮ��ƽ��ķ����жϣ��������Һ�е���غ㡢�����غ�ķ���Ӧ�ã����ջ����ǽ���ؼ�����Ŀ�Ѷ��еȣ�

��16H++10Z-+2XO4-�T2X2++5Z2+8H2O
��2A2++B2�T2A3++2B-
��2B-+Z2�TB2+2Z-
�ɴ��ƶ�����˵��������ǣ�������
| A�� | ��ӦZ2+2A2+�T2A3++2Z-���Խ��� | |
| B�� | ZԪ���ڷ�Ӧ���б���ԭ���ڷ�Ӧ���б����� | |
| C�� | ��������ǿ������˳����XO4-��Z2��B2��A3+ | |
| D�� | ��ԭ����ǿ������˳����Z-��B-��A2+��X2+ |
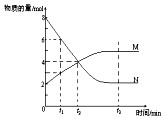 ��һ���¶��£�������ijһ��Ӧ��M��N�����ʵ����淴Ӧʱ��仯��������ͼ�����б�������ȷ���ǣ�������
��һ���¶��£�������ijһ��Ӧ��M��N�����ʵ����淴Ӧʱ��仯��������ͼ�����б�������ȷ���ǣ�������| A�� | ��Ӧ�Ļ�ѧ����ʽΪ��N�T2M | |
| B�� | t2ʱ����Ӧ����������Ũ����ȣ��ﵽƽ�� | |
| C�� | ��M��N��������ʱ����ѹǿ��N���ʵ������� | |
| D�� | �����¶ȣ�ƽ�ⳣ��K������Ӧ�ġ�H��0 |
| A�� | ij�л�����ȫȼ�����ɵ����ʵ�����CO2��H2O�����л���ķ���ʽһ��ΪCnH2n | |
| B�� | �춡���һ�ȴ�����2�� | |
| C�� | ��֬�������������������࣬������ͬϵ�� | |
| D�� | H2NCH2COOH�ȿ��Է���ȡ����Ӧ��Ҳ���Է������۷�Ӧ |
| A�� | ��ͭƬ����ϡ�����У�Cu+2H+�TCu2++H2�� | |
| B�� | �����۷����Ȼ�ͭ��Һ�У�Fe+Cu2+�TFe2++Cu | |
| C�� | Ba��OH��2��Һ�е���MgCl2��Һ�У�2OH-+Mg2+�TMg��OH��2�� | |
| D�� | �����еμ����CO32-+2H+�TH2O+CO2�� |
 pH=2��A��B��������Һ��1mL���ֱ��ˮϡ�͵�1000mL������pH����Һ���V�Ĺ�ϵ��ͼ��ʾ������˵����ȷ���ǣ�������
pH=2��A��B��������Һ��1mL���ֱ��ˮϡ�͵�1000mL������pH����Һ���V�Ĺ�ϵ��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | A��B������Һ�����ʵ���Ũ��һ����� | |
| B�� | ϡ�ͺ�A����Һ�����Ա�B����Һǿ | |
| C�� | a=5ʱ��A��ǿ�ᣬB������ | |
| D�� | ��aС��5����A��B�������ᣬ��A����������B |
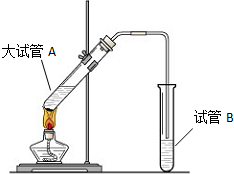 �����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺
�����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺