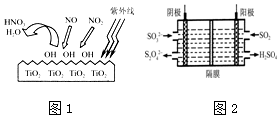
��Ŀ����
14�������л�ѧ��������������ƽ�ڲ�������ҩ�ע��ձ�����Ͳ������ƿ�߽�ͷ�ιܢ�ϸ���Լ�ƿ���ǩֽ
��1������Ҫ����480m L 1mol/L H2SO4��Һ����Ҫ��������Ϊ98%���ܶ�Ϊ1.84g/cm3��ŨH2SO427.2ml��
��2����ʵ�����������������������Һ�����ʵ���Ũ���к�Ӱ�죨�ƫ�ߡ���ƫ�͡����䡱��
������ϡ��Ũ������ձ�δϴ�ӣ�ƫ�ͣ�
��δ����ȴ����Һע������ƿ�У�ƫ�ߣ�
��ҡ�Ⱥ���Һ���½��ټ�ˮ��ƫ�ͣ�
�ܶ���ʱ�۲�Һ�温�ӣ�ƫ�ߣ�
����ȡŨ����ʱ���ӿ�Һ��ƫ�ߣ�
���� ��1���ȼ���Ũ��������ʵ���Ũ�ȣ��ٸ�����Һϡ��ǰ�����ʵ����ʵ��������������Ũ����������
��2���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��� �⣻��1����������Ϊ98%���ܶ�Ϊ1.84g/cm3��Ũ�������ʵ���Ũ��C=$\frac{1000��1.84��98%}{98}$=18.4mol/L������480m L 1mol/L H2SO4��Һ��Ӧѡ��500mL����ƿ������ҪŨ��������ΪV��1mol/L��500mL=18.4mol/L��V��V=27.2mL��
�ʴ�Ϊ��27.2��
��2��������ϡ��Ũ������ձ�δϴ�ӣ����²���������ģ����ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
��δ����ȴ����Һע������ƿ�У���ȴ��Һ���½�������Һ���ƫС����ҺŨ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
��ҡ�Ⱥ���Һ���½��ټ�ˮ��������Һ���ƫ����ҺŨ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
�ܶ���ʱ�۲�Һ�温�ӣ�������Һ���ƫС����ҺŨ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
����ȡŨ����ʱ���ӿ�Һ�棬������ȡ��Ũ�������ƫ����������ʵ���ƫ����ҺŨ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����Ƽ��й����ʵ���Ũ�ȼ��㣬��ȷ����ԭ�������������ǽ���ؼ���ע���������ķ�����

 ��ҵ�ϳɰ���ӦΪ��N2��g��+3H2��g�� $?_{���¸�ѹ}^{����}$ 2NH3��g���������о����£�
��ҵ�ϳɰ���ӦΪ��N2��g��+3H2��g�� $?_{���¸�ѹ}^{����}$ 2NH3��g���������о����£���1����֪H-H����Ϊ436kJ•mol-1��N-H����Ϊ391kJ•mol-1��N��N���ļ�����945.6kJ•mol-1����������Ӧ�ġ�H=-92.46kJ•mol-1��
��2��������Ӧ��ƽ�ⳣ��K�ı���ʽΪ��$\frac{{c}^{2}��N{H}_{3}��}{c��{N}_{2}��{c}^{3}��{H}_{2}��}$������Ӧ����ʽ��дΪ��$\frac{1}{2}$N2��g��+$\frac{3}{2}$H2��g��?NH3��g�����ڸ��¶��µ�ƽ�ⳣ����K1=$\sqrt{K}$����K��ʾ����
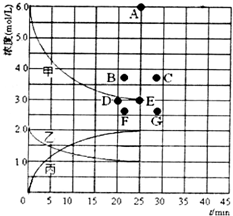
��3����773Kʱ���ֱ�2molN2��6molH2����һ���̶��ݻ�Ϊ1L���ܱ������У����ŷ�Ӧ�Ľ��У�����������n��H2����n��NH3���뷴Ӧʱ��t�Ĺ�ϵ���±���
| t/min | 0 | 5 | 10 | 15 | 20 | 25 | 30 |
| n��H2��/mol | 6.00 | 4.50 | 3.60 | 3.30 | 3.03 | 3.00 | 3.00 |
| n��NH3��/mol | 0 | 1.00 | 1.60 | 1.80 | 1.98 | 2.00 | 2.00 |
�����������е�ʵ�����ݼ���õ���Ũ�ȡ�ʱ�䡱�Ĺ�ϵ������ͼ�����߱�ʾ����ʾ c��N2����t���������ң��ڴ��¶��£�����ʼ����4molN2��12molH2����Ӧ�մﵽƽ��ʱ����ʾc��H2������������Ӧ�ĵ�ΪB��
| A�� | ��״���£�22.4 Lˮ�������ķ�����ԼΪ6.02��1023�� | |
| B�� | 1 mol Cl2�к��е�ԭ����ΪNA | |
| C�� | ��״���£�a L�����͵����Ļ���ﺬ�еķ�����ԼΪ$\frac{a}{22.4}$��6.02��1023�� | |
| D�� | ���³�ѹ�£�11.2 L CO��������0.5NA |
| A�� | ������ԭ����ͭ | |
| B�� | ������������ȼ�� | |
| C�� | Ba��OH��2•8H2O��NH4Cl��Ӧ | |
| D�� | ̼��Ƹ��·ֽ�������ƺͶ�����̼ |
| A�� | 2MnO2+2NH4++2e-�T2MnO��OH��+2NH3 | B�� | Zn-2e-�TZn2+ | ||
| C�� | [Zn��NH3��2]2++2e-�TZn+2NH3 | D�� | Mn4++e-�TMn3+ |
| A�� | ��������ѹǿһ����� | B�� | ��������A��Ũ��һ����� | ||
| C�� | ���������ջ�ų�������һ����� | D�� | ��������B�İٷֺ���һ����ͬ |
| A�� | ��ʼʱ�۲쵽������Ƭ�����м��������ݣ�����ԭ��Ϊ�ý���Ƭ����������Ĥ | |
| B�� | �����й۲쵽������Ƭ�����д������ݲ���Һ���ϡ���������˵���úϽ���Ƭ���ܶ�С�ڸ�������Һ���ܶ� | |
| C�� | ����������ʵ���Ũ��һ��Ϊ4.0 mol•L-1 | |
| D�� | ��ҺX��Mg2+��Al3+�����ʵ���֮��һ��Ϊ1��1 |
| A�� | ij����������Ϊ��2-��-3-�һ����� | |
| B�� | �ǻ��ĵ���ʽΪ��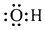 | |
| C�� | ������ӵ����ģ�ͣ� | |
| D�� | ����ļ���ʽ�� |