��Ŀ����
5��ij�����к�������CaCl2��Na2SO4��MgCl2Ϊ���ᴿҪ�����¹�������1������������ˮ��Ҫ���γ�ȥ���е�SO42-��Mg2+��Ca2+�����μ�����������Լ����������Լ�������ABC��������ţ�A��BaCl2 B��NaOH C��Na2CO3
��2�����������Na2CO3���������ӷ�ӦΪCa2++CO32-=CaCO3����Ba2++CO32-=BaCO3����
��3���������Ƶõľ����м���KIO3����ȡ���Σ�Ϊ�˼���ʳ�����Ƿ�KIO3�ɽ�ʳ������ˮ����������ữ��Ȼ�����KI���������·�Ӧ���м��飺KIO3+5KI+3H2SO4=3I2+3K2SO4+3H2O��������Ӧ���������ͻ�ԭ�����Ӹ����ı�Ϊ��1��5��
���� ��1�������е�SO42-�����Ȼ�����ȥ��Mg2+�������NaOH��ȥ��Ca2+��Na2CO3��Һ���ȿ��Գ�ȥ��Һ�еĸ����ӣ������Գ�ȥ��Һ�ж���ı����ӣ�
��2�����������Na2CO3ȥ�������ӵĶ���ı����ӣ��γ�̼�ᱵ��̼��Ƴ�����
��3����Ӧ��KIO3��IԪ�ػ��ϼ�Ϊ+5�ۣ�KI��IԪ�ػ��ϼ�Ϊ-1�ۣ���Ӧ��IԪ�ػ��ϼ۷ֱ���+5�ۡ�-1�۱仯Ϊ0�ۣ�KIO3Ϊ��������KIΪ��ԭ����
��� �⣺��1����ȥ�����еĿ��������ʣ�SO42-��Mg2+��Ca2+���������BaCl2��ȥ����������ӣ���SO42-+Ba2+=BaSO4�������Լ������NaOH��ȥ��þ���ӣ���Mg2++2OH-=Mg��OH��2�����������Na2CO3��ȥ�������ӵĶ���ı����ӣ���Ca2++CO32-=CaCO3����
�ʴ�Ϊ��ABC��
��2�����ε��ᴿ�У�����̼���Ƶ������dz�ȥ�������Ӹ������Լ������ı����ӣ���Ӧ�ķ���ʽΪ��CaCl2+Na2CO3=CaCO3��+2NaCl��BaCl2+Na2CO3=BaCO3��+2NaCl����Ӧ��ʵ���ǣ�Ca2++CO32-=CaCO3����Ba2++CO32-=BaCO3����
�ʴ�Ϊ��Ca2++CO32-=CaCO3����Ba2++CO32-=BaCO3����
��3����Ӧ��KIO3��IԪ�ػ��ϼ�Ϊ+5�ۣ�KI��IԪ�ػ��ϼ�Ϊ-1�ۣ���Ӧ��IԪ�ػ��ϼ۷ֱ���+5�ۡ�-1�۱仯Ϊ0�ۣ�KIO3Ϊ��������KIΪ��ԭ�����ɷ���ʽ��֪�������ͻ�ԭ�������ʵ���֮��Ϊ��KIO3����n��KI��=1��5��
�ʴ�Ϊ��1��5��
���� ���⿼�����йش��ε��ᴿ��������ԭ��Ӧ֪ʶ��������ѧ���ķ���������������ԭ��Ӧ��������Ŀ��飬ע��֪ʶ�Ĺ��ɺ������ǹؼ�����Ŀ�ѶȲ���

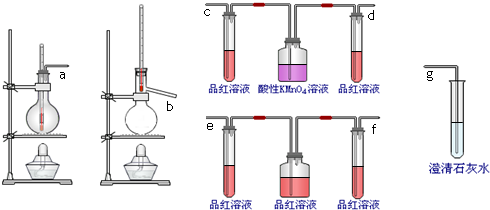
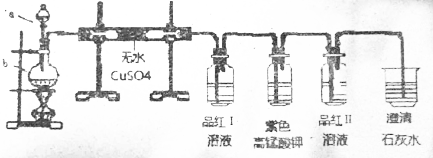
| A�� | ��ȡ����ˮʱ��Ϊ��ֹ��ƿ�ڲ�����������Ӧ������ƿ�м��뼸Ƭ���Ƭ | |
| B�� | ��������Ʊ�ʵ���У���β����Ⱦ������һ��Ҫ��ˮ����β�� | |
| C�� | �ü���KMnO4�ķ����Ƶõ������г�������ɫ��ʵ����Ӧ���Թܿڴ���һ���� | |
| D�� | �ڵ�ȼ��������ǰ��Ϊ��ֹ��ը��Ӧ���鴿 |
| A�� | HCl��Һ����OH- | |
| B�� | NaOH��Һ����H+ | |
| C�� | NaCl��Һ�м���OH-Ҳ��H+ | |
| D�� | �����£��κ����ʵ�ˮ��Һ�ж���H+��OH-����KW=c��H+��•c��OH-��=10-14 |
 ����ȩ��һ����Ҫ�Ļ���ԭ�ϣ�������ѧ�߶�ij��ѧ��ȤС������ʦ��ָ����������ͼװ�úϳ�����ȩ�������ķ�Ӧ���£�
����ȩ��һ����Ҫ�Ļ���ԭ�ϣ�������ѧ�߶�ij��ѧ��ȤС������ʦ��ָ����������ͼװ�úϳ�����ȩ�������ķ�Ӧ���£�CH3CH2CH2CH2OH$\underset{\stackrel{N{a}_{2}C{r}_{2}{O}_{7}}{��}}{{H}_{2}S{O}_{4}����}$CH3CH2CH2CHO
��Ӧ��Ͳ������������б����£�
| �е�/��c | �ܶ�/��g•cm-3�� | ˮ���ܽ��� | |
| ������ | 117.2 | 0.8109 | �� |
| ����ȩ | 75.7 | 0.8017 | �� |
��6.0gNa2Cr2O7����100mL�ձ��У���30mLˮ�ܽ⣬�ٻ�������5mLŨ���ᣬ��������ҺС��ת����B�У���A�м���4.0g�������ͼ�����ʯ�����ȣ�������������ʱ����ʼ�μ�B����Һ���μӹ����б��ַ�Ӧ�¶�Ϊ90-95�棬��E���ռ�90�����µ���֣�������ﵹ���Һ©���У���ȥˮ�㣬�л������������ռ�75-77����֣�����2.0g��
�ش��������⣺
��1��ʵ���У��ܷ�Na2Cr2O7��Һ�ӵ�Ũ�����У��������ɲ��ܣ�Ũ��������ˮ�ų������ȣ��������Ž����ˣ�
��2������װ��ͼ�У�D������������ֱ�������ܣ�E��������������ƿ��
��3��C1��C2�¶ȼ�ˮ����λ�ò�ͬ��ԭ����C1���Ʒ�Ӧ�¶ȣ�C2�ⶨ�������������¶�
��4����Ӧ�¶�Ӧ������90-95�棬��ԭ���DZ�֤����ȩ��ʱ��������ʹ��Ӧ������У��ֿɾ��������䱻��һ��������
��5����������ͬ���칹���������֣���д������������������������ͬ���칹�壺�ٺ�������������ͬ�Ĺ����ţ���һ���������ܱ�������ȩ����CH3��2CHCH2OH
��6����ʵ��������ȩ�IJ���Ϊ51%��
| A�� | ����AgNO3��Һ�����ɰ�ɫ��������ϡ����������ܽ�ʱ����ȷ����Cl-���� | |
| B�� | ͨ��Cl2����Һ��Ϊ���ɫ�����������Һ����Һ��������ȷ����I-���� | |
| C�� | ����BaCl2��Һ�����ɰ�ɫ��������ϡ�����������ܽ�ʱ����ȷ����SO42-���� | |
| D�� | ����NaOH��Һ�����ȣ����ɵ�������ʹ����ĺ�ɫʯ����ֽ��������ȷ����NH4+���� |