��Ŀ����
(1).���ڱ��ĺ�̼������Ȳ��ͬ��������Ϊ����һ�ֲ���������д��C6H6��һ�ֺ���������֧�������Ľṹ��ʽ______________________________________��
(2).������ʹ��ˮ��ɫ����������������д������������Ӧ�Ļ�ѧ����ʽ
____________________________________________________________________
 (3).��������ȥ2 mol��ԭ���γ�1 mol˫��Ҫ���ȣ���1��3��������ϩ��
(3).��������ȥ2 mol��ԭ���γ�1 mol˫��Ҫ���ȣ���1��3��������ϩ��![]() ��ȥ2 mol��ԭ�ӱ�ɱ�ȴ���ȣ����ƶϱ���1��3��������ϩ________�����ȶ����ȶ�����
��ȥ2 mol��ԭ�ӱ�ɱ�ȴ���ȣ����ƶϱ���1��3��������ϩ________�����ȶ����ȶ�����
(4).1866�꿭���գ���ͼ������˱��ĵ���˫���������������ƽ��ṹ�������˱��IJ������ʣ�������һЩ������δ����������ܽ������� ��ʵ�������ţ�___________
a.������ʹ��ˮ��ɫ b.������H2�����ӳɷ�Ӧ
c.�屽û��ͬ���칹�� d..�ڶ��屽ֻ��һ��
(5).�ִ���ѧ��Ϊ������̼̼֮��ļ���________________________________��
(1)![]() (���������𰸽Ը���)
(���������𰸽Ը���)
(2) ![]() + HNO3
+ HNO3 ![]()
![]() + H2O
+ H2O
��3���ȶ�
��4��ad
(5)����˫���͵���֮����صļ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
 ��
�� .
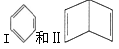
.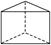 ���ýṹ�Ķ��ȴ�����________�֣�
���ýṹ�Ķ��ȴ�����________�֣�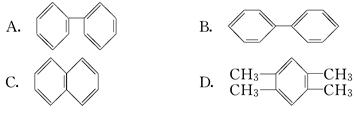
 �����ֽṹ��������������������棺���Է��棨����ѧ���ʷ��棩������_______������ĸ������I ���ܡ�
�����ֽṹ��������������������棺���Է��棨����ѧ���ʷ��棩������_______������ĸ������I ���ܡ� 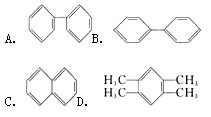
 )��ȥ2mol��ԭ��ת��ɱ�ȴ���ȣ����ƶϱ���1��3-������ϩ ____________����ȶ������ȶ�������
)��ȥ2mol��ԭ��ת��ɱ�ȴ���ȣ����ƶϱ���1��3-������ϩ ____________����ȶ������ȶ�������