��Ŀ����
��.��CH4����ԭNOx�������������������Ⱦ�����磺
CH4(g)��4NO2(g)===4NO(g)��CO2(g)��2H2O(g)��H����574 kJ��mol��1
CH4(g)��4NO(g)===2N2(g)��CO2(g)��2H2O(g)��H����1160 kJ��mol��1
����4.48 L(�ۺϳɱ�����) CH4��ԭNO2��N2������������ת�Ƶ�������Ϊ_________(�����ӵ�������ֵ��NA��ʾ)���ų�������Ϊ_________kJ��
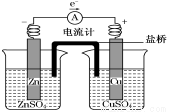
��.��1 L����0.4 mol CuSO4��0.2 mol NaCl ��ˮ��Һ�ö��Ե缫���һ��ʱ���
��1������һ���缫�ϵõ�0.1 mol Cu����һ�缫����������(�ڱ�״����)�������________L��
��2������һ���缫�ϵõ�0.4 mol Cu����һ�缫����������(�ڱ����)�������_________L��
��ϰ��ϵ�д�
 �����������Ů��ͯ������ϵ�д�
�����������Ů��ͯ������ϵ�д�
�����Ŀ


 CaO2��8H2O����Ӧʱͨ�����������Ca(OH)2����Ŀ����____________��
CaO2��8H2O����Ӧʱͨ�����������Ca(OH)2����Ŀ����____________��
 ��7H2O
��7H2O