��Ŀ����
����ʵ�鷽����ȷ���ǣ�������
| A������һ�����ʵ���Ũ�ȵ���Һʱ������ˮ��������ƿ�Ŀ̶��ߣ��ý�ͷ�ιܽ�����Һ���������� |
| B���ýྻ�IJ�˿պȡ������Һ�ھƾ��ƻ��������գ�����ʻ�ɫ������Һ��һ��������K+ |
| C������FeCl3��Һ��Fe��OH��3����ʱ���ɽ����Ƿֱ���һ�������䣬���������ЧӦ���� |
| D������ij��Һ���Ƿ���Fe3+ʱ�����ȼ�����������ˮ���ٵμ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+ |
���㣺���������ӵļ���,���ʵļ���ͼ���Ļ�������ѡ��Ӧ��,����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺
������A��������Һʱ����ˮ��������ƿ�Ŀ̶��ߣ�Ӧ�������ƣ�
B���ص���֤��Ӧ�ų��Ƶĸ��ţ�������ɫ�ܲ����۲죻
C��������ж����������Һ�����У�
D����֤���������Ƿ���ڣ����ô˷�����
B���ص���֤��Ӧ�ų��Ƶĸ��ţ�������ɫ�ܲ����۲죻
C��������ж����������Һ�����У�
D����֤���������Ƿ���ڣ����ô˷�����
���
�⣺A����Һ�����м�ˮ��������ƿ�Ŀ̶��ߣ���ʵ��ʧ�ܣ�Ӧ�������ƣ���A����
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ���������Һ�п��ܺ���K+����B����
C��������ж����������Һ�����У�������FeCl3��Һ��Fe��OH��3����ʱ���ɽ����Ƿֱ���һ�������䣬���������ЧӦ���֣���C��ȷ��
D�������ij��Һ���Ƿ���Fe3+ʱ���μ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+����D����
��ѡC��
B������ʻ�ɫ��һ���������ӣ��۲�K����ɫӦ����ɫ���ܲ���������Һ�п��ܺ���K+����B����
C��������ж����������Һ�����У�������FeCl3��Һ��Fe��OH��3����ʱ���ɽ����Ƿֱ���һ�������䣬���������ЧӦ���֣���C��ȷ��
D�������ij��Һ���Ƿ���Fe3+ʱ���μ����軯����Һ������Һ��Ϊ��ɫ����˵����Һ��һ������Fe3+����D����
��ѡC��
���������⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�漰��Һ���ơ����ʼ��𡢽������ʼ����Ӽ���ȣ�����ʵ��������������ʵĿ��飬ע�ⷽ���ĺ����ԡ������Է�����

��ϰ��ϵ�д�
�����Ŀ
��ӦC��s��+H2O��g��?CO��g��+H2��g����һ�ݻ��ɱ���ܱ������н��У����������ĸı���䷴Ӧ���ʼ�����Ӱ����ǣ�������
| A�����ܱ�������ͨ��ˮ���� |
| B������ |
| C������������䣬���뺤��ʹ��ϵѹǿ���� |
| D������ѹǿ���䣬���뺤��ʹ����������� |
��ǿ������Һ�У����и��������ܹ�����������ǣ�������
| A��Mg2+��Ca2+��HCO3-��CI- |
| B��Na+��CO32-��Cl-��SO42- |
| C��K+��Fe2+��SO42-��Br- |
| D��Fe2+��Ca2+��Cl-��NO3- |
����˵����ȷ���ǣ�������
| A��������0.4 mol/L HB��Һ��0.2 mol/L NaOH��Һ�������Ϻ���Һ��pH=3��������Һ������Ũ�ȵĴ�С˳��Ϊ��c��Na+����c��B-����c��H+����c��OH-�� |
| B������ʱ��pH=2��CH3COOH��Һ��HCl��Һ��pH=12�İ�ˮ��NaOH��Һ��������Һ����ˮ�����c��H+������� |
| C��pH=11��NaOH��Һ��pH=3�Ĵ�����Һ�������ϣ�����ʯ����Һ�ʺ�ɫ |
| D��0��lmol/L pHΪ4��NaHB��Һ�У�c��HB-����c��H2B����c��B2-�� |
��2.24g�����ۼ���50ml2mol?L-1���Ȼ�����Һ�У���ַ�Ӧ�������ǣ�������
| A������ʣ�࣬��Һ��dz��ɫ��Cl-Ũ�Ȼ������� |
| B������Һ�е���KSCN��ɫ��Һ�����Ժ�ɫ |
| C�����������뻹ԭ��������ʵ���֮��Ϊ2��5 |
| D��Fe2+��Fe3+�����ʵ���֮��Ϊ6��1 |
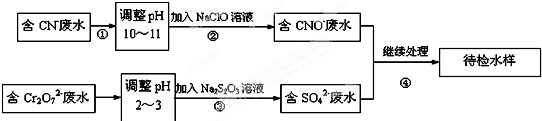
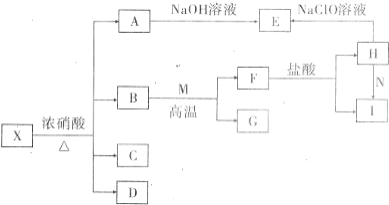 ����X�������ֳ���Ԫ����ɵĻ�������н���Ԫ������һԪ�ص�������Ϊ14��1����һ��������X�ɷ�������ת�������ֲ���δ�������C����ɫ��������ʹ����ʯ��ˮ����ǵ����壬DΪ����ɫ���壬EΪ���ɫ������MΪ�����������ʣ�
����X�������ֳ���Ԫ����ɵĻ�������н���Ԫ������һԪ�ص�������Ϊ14��1����һ��������X�ɷ�������ת�������ֲ���δ�������C����ɫ��������ʹ����ʯ��ˮ����ǵ����壬DΪ����ɫ���壬EΪ���ɫ������MΪ�����������ʣ�