��Ŀ����
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��Ը�����Ϊԭ���Ʊ��ظ����(K2C r2O7)�Ĺ�������(���ֲ�����������)��
r2O7)�Ĺ�������(���ֲ�����������)��
��.���������̼���ƻ�ϳ�ֱ��ա�
��.���պ�Ĺ����ˮ��ȡ������õ���ҺA����A��
��.����ҺA�м�������pHԼ7��8������õ���ҺB����B��
��.������ҺB�м����Ӵ����ữ��ʹ��ҺpHС��5��
��.��������Һ�м����Ȼ��أ��õ��ظ���ؾ��塣
(1)���б��շ����ķ�Ӧ���£���ƽ����д��ȱ��
FeO•Cr2O3 + Na2CO3 +  Na2CrO4 + Fe2O3 + CO2����
Na2CrO4 + Fe2O3 + CO2����
��Na2CO3+Al2O3=2NaAlO2+CO2����
(2)����A����Ҫ���� (��д��ѧʽ)��
(3)��֪�ظ������Һ�д�������ƽ�⣺2CrO42-��2H+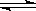 Cr2O72-��H2O�����е�����ҺpH��5ʱ����Ŀ���� ��
Cr2O72-��H2O�����е�����ҺpH��5ʱ����Ŀ���� ��
(4)���з�����Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl=K2Cr2O7��+2NaCl����֪�±�����
���� | KCl | NaCl | K2Cr2O7 | Na2Cr2O7 | |
�ܽ�� (g/100gˮ) | 0�� | 28[�� | 35.7 | 4.7 | 163 |
40�� | 40.1 | 36.4 | 26.3 | 215 | |
80�� | 51.3 | 38 | 73 | 376 | |
�ٸ÷�Ӧ�ܷ����������� ��
�ڻ��K2Cr2O7����IJ����жಽ��ɣ������ǣ�����KCl���塢 �� �����ˡ�ϴ�ӡ�����õ����塣
(5)���й���B����Ҫ��������������������þ���������ܻ����P���������ʣ���ȷ��������B���������������ķ����ǣ���ȡn g��Ʒ���������________(��д�Լ�����)���ܽ⡢���ˡ���ͨ�������CO2�������ա���ȴ���������ø������m g��������Ʒ��������������������Ϊ______ (�ú�m��n�Ĵ���ʽ��ʾ)��
 ��У����ϵ�д�
��У����ϵ�д����и��������У�������ͼ����һ��ת����ϵ��ѡ����

ѡ�� | X | Y | Z |
A | Na | NaOH | NaHCO3 |
B | Cu | CuSO4 | Cu(OH)2 |
C | C | CO | CO2 |
D | Si | SiO2 | H2SiO3 |



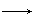 Mn2+ B��HCl
Mn2+ B��HCl O2
O2 -�������ӣ��������м��������Na2O2���Ȳ����裬��ͨ�������HI���壬��ַ�Ӧ�����
-�������ӣ��������м��������Na2O2���Ȳ����裬��ͨ�������HI���壬��ַ�Ӧ�����

 �ĵ���̶ȴ���ˮ��̶ȣ��ɲ��õ�ʵ�鷽����________(����ĸ)��
�ĵ���̶ȴ���ˮ��̶ȣ��ɲ��õ�ʵ�鷽����________(����ĸ)��
