��Ŀ����
3�������ҹ���ҵ��ˮƽ�IJ��Ϸ�չ�����ˮ��������Ⱦ�����Ϊ��Ҫ���⣮��1����ҵβ���к��д����ĵ������NH3����ԭ�������SCR��������ĿǰӦ����㷺���������������ѳ���������Ӧԭ����ͼ��ʾ��
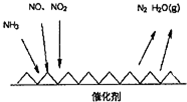
����ͼ��֪SCR�����е�������ΪNO��NO2
����Fe����������ʱ���ڰ�������������£��� NO2��NO�����ʵ���֮��Ϊ1��1ʱ��д���÷�Ӧ�Ļ�ѧ����ʽ2NH3+NO+NO2$\frac{Fe}{��}$2N2+3H2O��
��2����ҵ��ˮ�г�����һ������Cr2O72-��������༰��̬ϵͳ�����ܴ�����ⷨ�Ǵ�������Ⱦ�ij��÷������÷���Fe���缫��⺬Cr2O72-�����Է�ˮ�����ʱ�����������д����������ɣ�������Cr��OH��3��Fe��OH��3������
�ٷ�Ӧ��lmol Cr2O72-��ȫ����Cr��OH��3���������·ͨ�����ӵ����ʵ���Ϊ12mol��
�ڳ����£�Cr��OH��3���ܶȻ�Ksp=10-32����Cr3+Ũ��С��10-5mol•Lһ1��ʱ����Ϊ��ȫ�����������ȫ�����Һ��pH=6�������Һ���˺��ܣ���ܡ���ֱ���ŷţ�
��3��ClO2������һ�ֳ��õ������������ڱ��㷺����������ˮ��������������ˮ����ClO2�������ˮ�У�Ҫ��ClO2��Ũ����0.1��0.8mg•L-1֮�䣮���������Լ��ˮ��C102��Ũ�ȣ��������£�
I��ȡһ�������ˮ������������������Һ�������ԣ�Ȼ�����һ�����ĵ⻯�أ������������Һ����Һ������
����һ������Na2S2O3��Һ����֪��2S2O32-+I2=2S4O62-+2I-����
���������ˮ��pH��1.3��
��֪��������C1O2�����������»�ԭ����ΪClO3�������������»�ԭ����ΪC1-��
��ش��������⣺
��ȷ������II��ȫ��Ӧ��������ɫ��ʧ��������ڲ���ɫ
���ڲ���III�����У���Һ�ֳ���ɫ����Ӧ�����ӷ���ʽClO2-+4I-+4H+=Cl-+2I2+2H2O
����ˮ�������Ϊ1.0L���ڲ���IIʱ������1.0X 10-3mol•L-1��Na2S2O3��Һ10mL����ˮ����ClO2��Ũ����0.675mg•L-1��
���� ��1���ٵõ��ӣ����ϼ۽��͵ķ�Ӧ������������
�ڸ��ݻ��ϼ�����������ȡ�ԭ���غ�����ƽ��
��2�����������ӻ��Cr2O72-�����Է�ˮ��Ӧ��������Ӧ�����ӷ���ʽ��Cr2O72-+6Fe2++14H+�T2Cr3++6Fe3++7H2O����ϵ缫����ʽFe-2e-�TFe2+���㣻
�ڸ����ܶȻ����㣻
��3���ټ���һ���� Na2S2O3 ��Һ��������Ӧ2S2O32-+I2�TS4O62-+2I-��ʹI2��ԭΪI-��
����ͼʾ������pH��1��3ʱ��ClO2-��I-�������ɵ�I2����۽���ٴγ�����ɫ�����ݻ��ϼ�����������ȡ�ԭ���غ�͵���غ�����ƽ��
�۸��ݹ�ϵʽClO2��I-��S2O32-�ȼ����ClO2�����ʵ�����Ȼ���ټ����Ũ�ȣ�
��� �⣺��1������ͼ��֪SCR������NH3��NO��NO2��Ӧ����ΪN2��ˮ����������ΪNO��NO2��
�ʴ�Ϊ��NO��NO2��
��NH3�е��Ļ��ϼ���-3�����ߵ�0�ۣ�һ��NH3ʧȥ3�����ӣ�NO2�е��Ļ��ϼ���+4�۽��͵�0�ۣ�һ��NO2�õ�4�����ӣ�NO�е��Ļ��ϼ���+2�۽��͵�0�ۣ�һ��NO�õ�2�����ӣ���NO2��NO�����ʵ���֮��Ϊ1��1ʱ��ת�Ƶ��ӵ���С������Ϊ6����������غ��֪����ʽΪ��2NH3+NO+NO2$\frac{Fe}{��}$2N2+3H2O��
�ʴ�Ϊ��2NH3+NO+NO2$\frac{Fe}{��}$2N2+3H2O��
��2�����������ӻ��Cr2O72-�����Է�ˮ��Ӧ��������Ӧ�����ӷ���ʽ��Cr2O72-+6Fe2++14H+�T2Cr3++6Fe3++7H2O�������缫����ʽFe-2e-�TFe2+����
Cr2O72-��6Fe2+��12mol���ӣ�1mol Cr2O72-��ȫ����Cr��0H��3���������·ͨ�����ӵ����ʵ���Ϊ12mol��
�ʴ�Ϊ��12��
����Һ��pH=6����c��OH-��=10-8mol•L-1����c3��OH-����c��Cr3+��=Ksp=10-32��c��Cr3+��=10-8mol•L-1��10-5mol•L-1����ֱ���ŷţ�
�ʴ�Ϊ���ܣ�
��3���ټ���һ���� Na2S2O3 ��Һ��������ӦΪ��2S2O32-+I2�TS4O62-+2I-��ʹI2��ԭΪI-������ɫ����ʧ���ʴ�Ϊ����ɫ��ʧ��������ڲ���ɫ��
����ͼʾ������pH��1��3ʱ��ClO2-��I-�������ɵ�I2��ClO2-���ȵĻ��ϼ���+3�۽��͵�-1�ۣ�һ��ClO2-�õ�4�����ӣ���Ļ��ϼ���-1�����ߵ�0�ۣ�һ��I-ʧȥ1�����ӣ�ת�Ƶ��ӵ���С������Ϊ4����������غ㡢����غ��֪���ӷ���ʽΪ��ClO2-+4I-+4H+=Cl-+2I2+2H2O��
�ʴ�Ϊ��ClO2-+4I-+4H+=Cl-+2I2+2H2O��
��ClO2 ��I-��S2O32-
1 1
1.0��10-5mol 1.0��10-3 mol/L��0.01L
m��ClO2��=n��ClO2����M��ClO2��=1.0��10-5mol��67.5��103mg•mol-1=0.675mg
����ˮ�������Ϊ1.0L������ ClO2 ��Ũ��Ϊ$\frac{0.675mg}{1L}$=0.675 mg•L-1��
�ʴ�Ϊ��0.675��
���� ������Ҫ������������ԭ��Ӧ�����ӷ���ʽʽ����д�Լ���ѧ���㣬�Ѷ��еȣ�ע������ʵ��ԭ���͵��ӵ�ʧ��ƽ�ķ�����

 ���źþ���Ԫ����ĩ��ϵ�д�
���źþ���Ԫ����ĩ��ϵ�д�| A�� | 1molCl2�����������۷�Ӧ����Ӧת�Ƶ��ӵ���ĿΪ3NA | |
| B�� | ����ʱ������Zn��Ũ���ᷴӦ������2mol���壬ת�Ƶ�����Ϊ4NA | |
| C�� | 7.8gNa2O2������ˮ��Ӧʱת�Ƶ�����ĿΪ0.2NA | |
| D�� | 1mol��������������ú��ڱ�״���µ����Ϊ22.4L |
| A�� | 1 | B�� | l-a | C�� | 2-lga | D�� | 3-lga |
| A�� | 50mL18.4mol/LŨ����������ͭ�ȷ�Ӧ������SO2���ӵ���ĿΪ0.46NA | |
| B�� | ij�ܱ�����ʢ��0.1mol N2��0.3mol H2����һ�������³�ַ�Ӧ��ת�Ƶ��ӵ���ĿΪ0 6NA | |
| C�� | ����CO2��O2��ɵĻ�����й���NA�����ӣ������е���ԭ����Ϊ2 NA | |
| D�� | �����£�1L 0�� 1 mol/L NH4NO3��Һ�е���ԭ����Ϊ0.4 NA |
| A�� | ԭ�Ӱ뾶��W��Z��Y | |
| B�� | ��ҵ��ͨ�����Y����ˮ�����Ʊ�Y�ĵ��� | |
| C�� | ����������Ӧˮ��������ԣ�W��Z��X | |
| D�� | ��Ԫ�طֱ���X��Y��Z��W�γɵĻ����ﶼ��ֹһ�� |