��Ŀ����
3�����ʵ���Ũ����ͬ�����и������ʵ���Һ�У���ָ�����ӵ�Ũ������С�Ƚϣ����д�����ǣ�������| A�� | c��PO43-����Na3PO4��Na2HPO4��NaH2PO4��H3PO4 | |
| B�� | c��CO32-������NH4��2CO3��Na2CO3��NaHCO3��NH4HCO3 | |
| C�� | c��NH4+������NH4��2SO4����NH4��2CO3��NH4HSO4��NH4Cl | |
| D�� | c��S2-����Na2S��NaHS��H2S |
���� A����������ߵ������c��H+�������Բ���PO43-�ĵ������������ã�
B��̼������̼�������Ũ�ȴ���̼���������Ũ�ȣ�Ȼ������ε�ˮ��Ӱ���ж�̼�������Ũ�ȴ�С��
C����������狀�̼����е�笠�����Ũ�ȴ�����ʽ����笠�����Ũ�ȣ����������������������笠����ӵĵ��룻
D��������������Ũ����������Ƕ�Ԫ���ᣬ������Ũ����С��
��� �⣺A��Na3PO4�����������Ũ�����Na2HPO4��NaH2PO4��H3PO4�������c��H+�������������ӶԲ���PO43-�ĵ������������ã���Һ�����������Ũ�ȴ�СΪ��Na3PO4��Na2HPO4��NaH2PO4��H3PO4����A��ȷ��
B��Na2CO3�������ǣ�NH4��2CO3����Ϊ����Ҫ����ˮ�⣬NaHCO3��NH4HCO3����HCO3-�����������NH4HCO3��HCO3-��NH4+��ٽ�ˮ�⣬HCO3-Ũ�Ƚ�С��NaHCO3��NH4HCO3����B����
C�������ξ���ȫ���룬��NH4��2SO4 �ͣ�NH4��2CO3�ϴ����ߵ��������ӻᷢ����ٽ���ˮ�⣬ӦΪ��NH4��2SO4����NH4��2CO3��NH4HSO4��NH4Cl��NH4HSO4 ���������H+��NH4+��ˮ�����������ã�ӦΪNH4HSO4��NH4Cl����Һ��笠�����Ũ�ȴ�СΪ����NH4��2SO4����NH4��2CO3��NH4HSO4��NH4Cl����C��ȷ��
D��Na2S���H2S��NaHS��ȣ���ǰ����������������������ӵĵ��룬������Ũ��ӦΪNaHS��H2S����Һ��������Ũ�ȴ�СΪ��Na2S��NaHS��H2S����D��ȷ��
��ѡB��
���� ���⿼��������Ũ�ȴ�С�Ƚϣ���Ŀ�Ѷ��еȣ���ȷ�ε�ˮ��ԭ������Ӱ��Ϊ���ؼ���ע������������ʵĵ���ƽ�⼰��Ӱ�죬����������ѧ���ķ������������Ӧ��������

 С��ſ�ʱ��ҵϵ�д�
С��ſ�ʱ��ҵϵ�д� һ������ϵ�д�
һ������ϵ�д� �Ƹ�С״Ԫ���ֳ������ϵ�д�
�Ƹ�С״Ԫ���ֳ������ϵ�д� �¸��̵�ѧϵ�д�
�¸��̵�ѧϵ�д� ����ͬѧһ����ʦȫ�źþ�ϵ�д�
����ͬѧһ����ʦȫ�źþ�ϵ�д�| A�� | ���³�ѹ�£�11.2 L�����к��е���ԭ����Ϊ2NA | |
| B�� | ��״���£�0.3 mol������̼�к�����ԭ����Ϊ0.3 NA | |
| C�� | �����£�2.7 g�������������ᷴӦ��ʧȥ�ĵ�����0.3 NA | |
| D�� | �����£�0.1 mol/L MgCl2��Һ�к�Cl-��Ϊ0.2 NA |
| A�� | �����£�ŨH2SO4�����������������������棬˵��ŨH2SO4��Al����Fe�������²���Ӧ | |
| B�� | �������м���ŨH2SO4����ַ�������˵��ŨH2SO4������ˮ�� | |
| C�� | ŨH2SO4��ʹ����������ף�˵��ŨH2SO4������ˮ�� | |
| D�� | ϡH2SO4����Fe��Ӧ����H2��˵��ϡH2SO4Ҳ�������� |
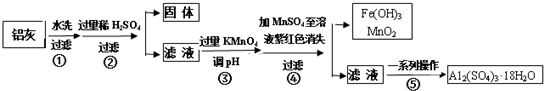
��ش��������⣺
��1���������ϡH2SO4�ܽ�A12O3�����ӷ���ʽ��6H++Al2O3=2Al3++3H2O��
��2�������м����KMnO4Ҳ����H2O2���棬����H2O2������Ӧ�Ļ�ѧ����ʽΪH2O2+2FeSO4+H2SO4=Fe2��SO4��3+2H2O��
��3����֪��Ũ�Ⱦ�Ϊ0.1mol/L�Ľ��������ӣ������������������pH�����
| Al��OH��3 | Fe��OH��2 | Fe��OH��3 | |
| ��ʼ����ʱ | 3.4 | 6.3 | 1.5 |
| ��ȫ����ʱ | 4.7 | 8.3 | 2.8 |
��4����֪Ksp[Fe��OH��3]=c��Fe3+��•c3��OH-��=4.0��10-38�������£���pH=2ʱ��Fe3+��ʼ������Ũ��Ϊ4.0��10-2mol/L��
��5�������ܷ�����Ӧ�����ӷ���ʽΪ2MnO4-+3Mn2++2H2O=5MnO2+4H+��Ϊ����֤�ò������ù�����ȷʵ����MnO2����ѡ�õ��Լ���Ũ�����˫��ˮ��
��6�������ݡ�һϵ�в������������������ò�������B ������ţ���
A�������� B������ C�������� D���ƾ��� E��©����
| �� | �� | |
| �� | �������� | �����ӵ����� |
| �� | ���������Ħ����� | ������������ |
| �� | �ܼ������ | ���ʵ����ʵ���Ũ�� |
| �� | ��Һ�����ʵ��������� | ��Һ������ |
| �� | �DZ�������ʵ����� | ���ʵ�Ħ������ |
| A�� | �� | B�� | �ۢ� | C�� | �ڢۢ� | D�� | �ۢܢ� |
| A�� | ������ԭ�ӵĸ�����Ϊ2��3 | B�� | ������ԭ�ӵĸ�����Ϊ1��1 | ||
| C�� | ������Ԫ�ص�������Ϊ5��6 | D�� | ������Ԫ�ص�������Ϊ5��4 |
 ���ʽṹ�����������ʣ��ش��������⣺
���ʽṹ�����������ʣ��ش��������⣺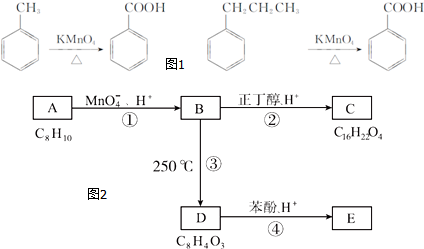
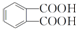 ��
��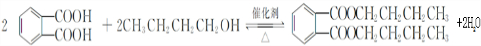 ���÷�Ӧ������Ϊȡ����Ӧ��
���÷�Ӧ������Ϊȡ����Ӧ��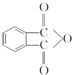 ����D���ʵĺ˴Ź�������ͼ�У������2��壬�����֮��Ϊ1��1��
����D���ʵĺ˴Ź�������ͼ�У������2��壬�����֮��Ϊ1��1��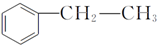 ��д�ṹ��ʽ����
��д�ṹ��ʽ����