��Ŀ����
��1����14.2g��ˮ���������Ƴ�500mL��Һ�������ʵ���Ũ��Ϊ mol/L��
��2��������ȡ��50mL�������ʵ���Ũ��Ϊ mol/L�����ʵ�����Ϊ g��
��3��������50mL��Һ��ˮϡ�͵�100mL��������Һ��Na+�����ʵ���Ũ��Ϊ mol/L��SO42-�����ʵ���Ũ��Ϊ mol/L��
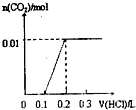
��4����״���µ�67.2LHCl���庬�е�HCl�����ʵ���Ϊ mol��������Ϊ ��HCl������Ϊ g����ͬ���ʵ����ĸ���������ˮ�Ƴ�500ml��Һ�����������ʵ���Ũ��Ϊ ��
��2��������ȡ��50mL�������ʵ���Ũ��Ϊ
��3��������50mL��Һ��ˮϡ�͵�100mL��������Һ��Na+�����ʵ���Ũ��Ϊ
��4����״���µ�67.2LHCl���庬�е�HCl�����ʵ���Ϊ
���㣺���ʵ���Ũ�ȵ���ؼ���,���ʵ�������ؼ���
ר�⣺������
��������1���ȼ��������Ƶ����ʵ������ٸ������ʵ���Ũ�ȹ�ʽ������Һ�����ʵ���Ũ�ȣ�
��2��������ҺŨ�ȵľ�һ�ԣ���ȡ��ҺŨ����ԭ��ҺŨ��һ��������ȡ��Һ�������С�أ��Ӷ��ж�50mL��Һ��Ũ�ȣ�ͬһ��Һ�����ʵ�Ũ����ͬ�����ʵ���������Һ����������ȣ��ݴ˼������ʵ�������
��3��������Һϡ��ǰ�����ʵ����ʵ����������ϡ�ͺ���Һ�����ʵ���Ũ�ȣ�Ȼ��������ʵķ���ʽ�����ӵĹ�ϵ�������ӵ�Ũ�ȣ�
��4������n=
����������67.2LHCl�����ʵ������ٸ���N=nNA��������еķ�������Ȼ�����m=nM������Ȼ����������������c=
�������Һ��Ũ�ȣ�
��2��������ҺŨ�ȵľ�һ�ԣ���ȡ��ҺŨ����ԭ��ҺŨ��һ��������ȡ��Һ�������С�أ��Ӷ��ж�50mL��Һ��Ũ�ȣ�ͬһ��Һ�����ʵ�Ũ����ͬ�����ʵ���������Һ����������ȣ��ݴ˼������ʵ�������
��3��������Һϡ��ǰ�����ʵ����ʵ����������ϡ�ͺ���Һ�����ʵ���Ũ�ȣ�Ȼ��������ʵķ���ʽ�����ӵĹ�ϵ�������ӵ�Ũ�ȣ�
��4������n=
| V |
| 22.4L/mol |
| n |
| V |
���
�⣺��1�������Ƶ����ʵ�����n=
=0.1mol������������ʵ���Ũ��Ϊ��c=
=0.2mol/L���ʴ�Ϊ��0.2��
��2��������ҺŨ�ȵľ�һ�ԣ���ȡ��ҺŨ����ԭ��ҺŨ��һ��������ȡ��Һ�������С�أ�
��ҺŨ����ͬ�����ʵ���������ȡ��Һ����������ȣ���50mL��Һ�����ʵ�����Ϊxg��
���Ա���ʽΪ��14.2g��xg=0.5L��0.05L����ã�x=1.42��
�ʴ�Ϊ��0.2��1.42��
��3����Һϡ��ǰ�����ʵ����ʵ�������ã�c1V1=c2V2��c2=
=
=0.1mol/L��
���������ƵĻ�ѧʽ��֪��Na+�����ʵ���Ũ��Ϊ0.2mol/L��SO42-�����ʵ���Ũ���������Ƶ����ʵ���Ũ����ȣ�Ϊ0.1mol/L��
�ʴ�Ϊ��0.2��0.1��
��4����״���µ�67.2LHCl���庬�е�HCl�����ʵ���Ϊ��n=
=3mol�������Ȼ���ķ�����Ϊ3NA��
3mol�Ȼ��������Ϊ��36.5g/mol��3mol=109.5g��
��ͬ���ʵ����ĸ���������ˮ�Ƴ�500mL��Һ��������ҺŨ��Ϊ��c=
=6mol/L��
�ʴ�Ϊ��3��3NA��109.5��6mol/L��
| 14.2g |
| 142g/mol |
| 0.1mol |
| 0.5L |
��2��������ҺŨ�ȵľ�һ�ԣ���ȡ��ҺŨ����ԭ��ҺŨ��һ��������ȡ��Һ�������С�أ�
��ҺŨ����ͬ�����ʵ���������ȡ��Һ����������ȣ���50mL��Һ�����ʵ�����Ϊxg��
���Ա���ʽΪ��14.2g��xg=0.5L��0.05L����ã�x=1.42��
�ʴ�Ϊ��0.2��1.42��
��3����Һϡ��ǰ�����ʵ����ʵ�������ã�c1V1=c2V2��c2=
| c1V1 |
| V2 |
| 0.2mol/L��0.05L |
| 0.1L |
���������ƵĻ�ѧʽ��֪��Na+�����ʵ���Ũ��Ϊ0.2mol/L��SO42-�����ʵ���Ũ���������Ƶ����ʵ���Ũ����ȣ�Ϊ0.1mol/L��
�ʴ�Ϊ��0.2��0.1��
��4����״���µ�67.2LHCl���庬�е�HCl�����ʵ���Ϊ��n=
| 67.2L |
| 22.4L/mol |
3mol�Ȼ��������Ϊ��36.5g/mol��3mol=109.5g��
��ͬ���ʵ����ĸ���������ˮ�Ƴ�500mL��Һ��������ҺŨ��Ϊ��c=
| 3mol |
| 0.5L |
�ʴ�Ϊ��3��3NA��109.5��6mol/L��
���������⿼�������ʵ���Ũ�ȵ���ؼ��㣬��Ŀ�Ѷ��еȣ�ע���������ʵ���Ũ�ȵĸ����ȷ���ʵ�����Ħ�����������ʵ���Ũ�ȵ�������֮���ת����ϵΪ���ؼ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
ij��ѧ����С���о���������������ʱ���ı�ijһ�����Է�Ӧ[����aA��s��+bB��g��?cC��g����ʾ]�Ļ�ѧƽ���Ӱ�죬�õ���ͼͼ��ͼ��p��ʾѹǿ��T��ʾ�¶ȣ�n��ʾ���ʵ���������ʾƽ��ת���ʣ�?��ʾ���������������ͼ�������ж���ȷ���ǣ�������
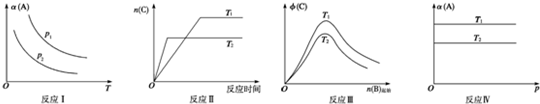

| A����Ӧ����p1��p2����a+b��c |
| B����Ӧ�˷�Ӧ�ġ�H��0����T1��T2 |
| C����Ӧ��H��0��T2��T1���H��0��T2��T1 |
| D����Ӧ����T1��T2����÷�Ӧ�����Է����� |
ǿ����ǿ���ϡ��Һ�����кͷ�Ӧ����ЧӦ��H+��aq��+OH- ��aq��=H2O��l����H=-57.3KJ/mol����1L 0.5mol/L��NaOH��Һ�м���ϡ���ᡢŨ���ᡢϡ���ᣬ��ǡ����ȫ��Ӧʱ����ЧӦ��H1����H2����H3�Ĺ�ϵ��ȷ���ǣ�������
| A����H1����H2����H3 |
| B����H1����H3����H2 |
| C����H2����H1����H3 |
| D����H1����H3����H2 |
��-235����Ԫ�ص�һ�ַ����Ժ��أ����й���
U��˵����ȷ���ǣ�������
235 92 |
| A��������Ϊ92 |
| B��������Ϊ143 |
| C�����������Ϊ143 |
| D��������Ϊ235 |
���и������ʵ�ȼ������ȵ��ǣ�������
| A��̼��һ����̼ |
| B��1 mol̼��3 mol̼ |
| C��3 mol C2H2��1 mol C6H6 |
| D�����ʯ��ʯī |
����˵����ȷ���ǣ�������
| A��1 mol O��������16 g/mol |
| B��Na��Ħ��������23 g/mol |
| C��CO2��Ħ��������44 g |
| D�����Ħ��������2 g/mol |
�������������ӷ���ʽ��д��ȷ���ǣ�������
| A����������������ͨ�����������Һ�У�SO2+H2O+ClO-=SO42-+Cl-+2H+ | ||||
| B������Fe2O3����Һ��ͨ��HI���壺Fe2O3+6H+=2Fe3++3H2O | ||||
C���ö��Ե缫���MgCl2��Һ��2Cl-+2H2O
| ||||
| D�������ʵ�����Ba��OH��2�루NH4��2Fe��SO4��2����Һ�з�Ӧ��Ba2++2OH-+2NH4++SO42-=BaSO4��+2NH3?H2O |